Earth and Life Science Reviewer (With Free Practice Tests)

Earth science studies the dynamic Earth and its processes, properties, structures, and relationship with its neighbors in space.
While it is composed of different sciences, it can be condensed down into four main disciplines: geology (the study of earth materials), meteorology (the study of the atmosphere), oceanography (the study of oceans), and astronomy (the study of celestial bodies).
In this review, we will be delving into the different processes and systems that shape the Earth and how they affect us.
Click below to go to the main reviewer:
Table of Contents
1. Earth’s Vital Statistics and Earth Systems
According to the data from NASA, the Earth’s vital statistics are as follows:
- Age: 4.543 billion years old
- Equatorial circumference: 40,075 km
- Equatorial radius: 6,378 km
- Polar radius: 6,356 km
- Total mass: 5.972 x 1024 kg
- Total volume: 1.08 x 1012 km3
- Total surface area: 5.10 x 108 km2
- Average density: 5.513 g/cm3
The Earth is composed of systems or “spheres”, each having their unique properties, that continuously interact with one another. We can divide them into four major spheres
a. Atmosphere
The atmosphere is a collective layer of gas that envelopes the Earth. It can be further divided into different layers based on characteristics.
The atmosphere is essential to life on Earth because (1) It shields the Earth and its inhabitants from harmful ultraviolet (UV) radiation from the Sun; (2) it maintains the warmth of the Earth’s surface; and (3) it contains all of the essential gases needed to support life. More about the atmosphere will be covered later on.
b. Hydrosphere
The hydrosphere refers to the bodies of water consisting of freely flowing bodies of water found on the surface of the Earth, as well as water reservoirs stored below the ground as groundwater.
This sphere covers nearly 71% of the Earth’s surface.
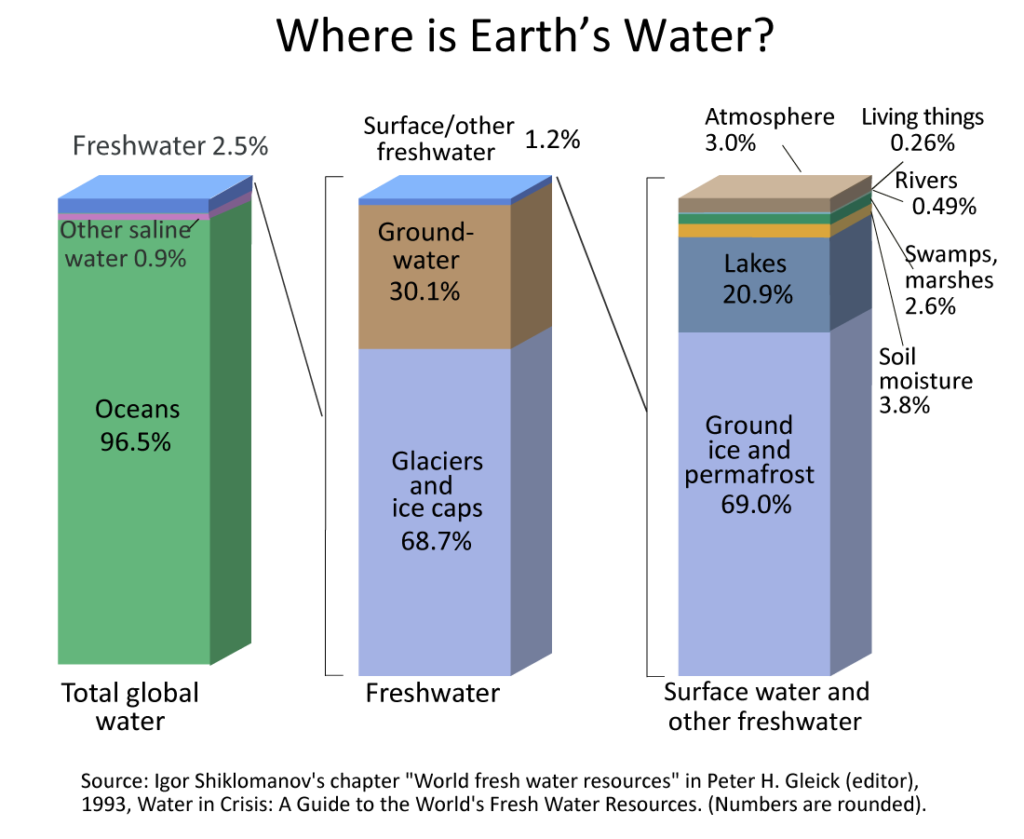
Nearly 97.4% of the water in the hydrosphere is composed of saline or salt water found in vast oceans covering the Earth. The remaining 2.6% comprises fresh water stored in glaciers, ice caps, and underground reservoirs.
Although freshwater makes up only a tiny fraction of the total, we and other Earth residents rely on it for survival.
c. Biosphere
The biosphere refers to the narrow band on the Earth’s surface where all biological life resides. This could range from bustling cities, lush tropical rainforests, arid deserts, or even extreme environments like the bottom of the ocean floor.
d. Geosphere
The geosphere is the largest out of all the spheres, extending from the surface of the Earth down to its center. It comprises external processes that we can observe on the surface and internal processes deep within.
Here’s a fun fact: soil can be considered the interface of the four spheres. It is made up of weathered or broken down rock (geosphere), organic matter or humus (biosphere), moisture (hydrosphere), and air (atmosphere).
Module 1 Practice Test
2. The Layers of the Earth and Its Composition
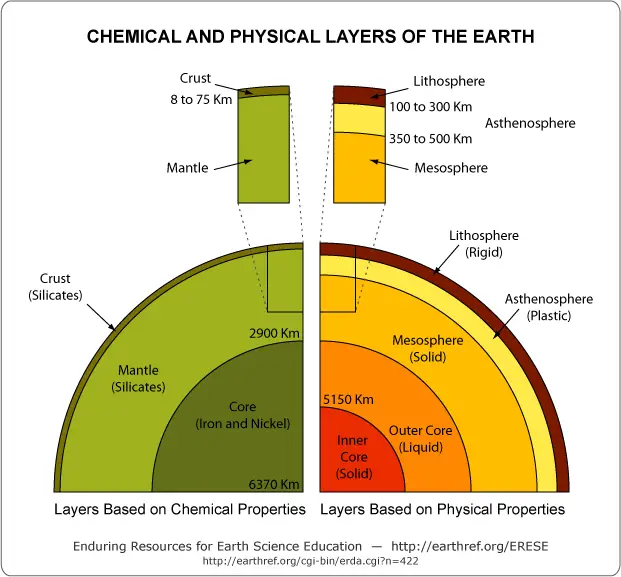
The Earth can be subdivided into layers based on two criteria: (1) composition (density) differences and (2) physical properties.
a. Based on Compositional Differences
i. Crust
This is the thinnest and outermost layer of the Earth. There are two types of crust– the continental and oceanic crust.
The continental crust is the older and more buoyant type of crust. It has an average thickness of 35 km but can be more than 70 km thick in mountainous regions. It has an average composition of granite with a 2.7 g/cm3 density.
The oceanic crust is the younger and denser type of crust. It has an average thickness of 7 km, much thinner than the continental crust. It comprises a 3.0 g/cm3 density of basalt, dark igneous rock.
ii. Mantle
The mantle comprises most of the Earth’s volume (more than 80%) and begins where the crust ends, down to a depth of 2,900 km.
The boundary between the crust and mantle is called the Mohorovičić discontinuity, marked by a chemical composition change.
It can be divided into the upper and lower mantle, separated by the Repetti discontinuity.
iii. Core
The core begins at the mantle-core boundary, the Gutenberg discontinuity, located at the 2,900 km depth.
Although no one has ever been to or sampled the core, scientific investigations concluded that its composition comprises a Fe-Ni (iron and nickel) alloy.
Due to its composition and the pressure conditions at depth, it is calculated to have a whopping density of around 11 g/cm3.
b. Based on Physical Properties
i. Lithosphere
The lithosphere (the Greek word lithos meaning “stone”) is a thick and brittle layer comprising the entire crust and uppermost layer of the upper mantle.
It has an average thickness of 100 km but can reach up to 300 km in the thickest portions of continents.
ii. Asthenosphere
The asthenosphere (from the Greek word asthenēs meaning “weak”) is a mechanically weak layer consisting of the lower portion of the upper mantle, extending to 660 km.
Contrary to popular belief, it is not a “sea of molten rock.” The upper mantle comprises an Mg- and Fe-rich rock called peridotite.
At this depth, the temperature and pressure conditions are high enough that rocks become ductile and deform easily. Because of this, the asthenosphere flows more like very viscous fluid (but remember: it is not liquid!) and moves independently from the overlying lithosphere. This is a crucial mechanism for plate tectonics (which will be discussed later in more detail).
iii. Mesosphere
Beneath the asthenosphere is the mesosphere (from the Greek word mesos meaning “middle”), comprised of the lower mantle, reaching the 2,900 km depth.
The dominant rock type in this layer is a silicate rock called perovskite.
Unlike the asthenosphere, the mesosphere is much stronger and flows with more resistance. Because of the immense pressure from the overlying layers, the strength of this layer increases with depth.
iv. Outer Core
Unlike all the other mechanical layers, the outer core is the only one made out of liquid–melted Fe-Ni alloy, to be exact.
The liquid nature of this layer can be attributed to extremely high temperatures (more than 3000°C!) that melt Fe, Ni, and all other elements.
The flow of liquid metals is responsible for the Earth’s magnetic field. The outer core terminates at a depth of 5,150 km, where the solid inner core begins. The outer-inner core boundary is also known as the Lehmann discontinuity.
v. Inner Core
Despite the extreme temperature, the overwhelming pressure in this layer forces the inner core to be a solid ball of mostly Fe.
Temperatures in the inner core are similar to the temperatures of the surface of the Sun—around more than 5400°C.
Module 2 Practice Test
3. Minerals
Minerals are building blocks of rocks. To be considered a mineral, it must be the following:
- Naturally-occurring. Man-made materials such as synthetic diamonds cannot be considered real minerals.
- Inorganic. Organic materials such as pearls or sugar are not minerals.
- Homogeneous solid. Minerals should be crystalline solids. Water is not a mineral, whereas ice is considered a mineral. Mercury occurs as a liquid in its natural state and is regarded as a mineraloid.
- Has definite chemical composition. You should be able to describe a mineral’s composition using a chemical formula.
- Ordered crystalline structure. Atoms in a mineral are placed in a repetitive and orderly manner. Substances that lack this kind of atomic structure, such as obsidian (volcanic glass) or plastic, are not considered minerals.
Properties of Minerals
Minerals come in all sorts of appearances. To identify different minerals, certain properties are observed in hand specimens. Here are some of the most commonly used properties in describing a mineral’s appearance:
- Color. It refers to the wavelengths of light reflected by the minerals. While it can be tempting to identify a mineral based on its color, it is the least valuable property because many minerals can occur in different colors.
- Luster. It describes how light is reflected from the mineral’s surface. A mineral could have a metallic luster or nonmetallic luster similar to pearls (pearly), glass (vitreous), resin (resinous), silk (silky), or others. Brilliantly cut gems are described to have an adamantine luster.
- Crystal Habit or Shape. This refers to the shape of each crystal or an aggregate of crystals. Although a single mineral can occur in various shapes, crystal habit can still be an identifying feature in certain minerals.
- Streak. This is the color of the mineral when it is powdered. Some minerals have different streak colors than their apparent color, which becomes a useful property when differentiating similar-looking minerals.
- Hardness. This refers to how resistant a mineral is to scratching. The Mohs’ Hardness Scale is a tool used to describe a mineral’s hardness relative to other minerals.

- Cleavage or Fracture. Cleavage refers to the tendency of a mineral to break along preferred planes called zones of weakness. A fracture is produced if a mineral doesn’t break along zones of weakness.
- Density or Specific Gravity. This refers to the ratio between a mineral’s weight and the weight of a specific volume of water (Water has a specific gravity of 1). Heavy minerals such as gold or platinum have very high specific gravity, whereas light minerals such as graphite have low specific gravity.
- Tenacity. This describes how well a mineral handles stress, such as breaking, crushing, bending, or tearing. Minerals susceptible to cracking or breaking are called brittle (e.g., quartz, calcite). A mineral that deforms under stress but snaps back to its original shape after the pressure is removed is called elastic (e.g., mica minerals). On the other hand, if a mineral is deformed under stress but doesn’t go back to its original shape, it is then called flexible (e.g., vermiculite). Metallic minerals such as gold, copper, or silver are called malleable because they can be flattened into sheets. Copper is also ductile because it can be drawn into thin wires without breaking. Sectile minerals such as gold or gypsum can be carved into thin sheets with a knife.
- Diaphaneity. This refers to how well light travels through a mineral. Transparent minerals allow almost all light to travel through the mineral (e.g., some quartz and calcite). Translucent minerals only allow some light to travel and exit the mineral, giving off a cloudy or murky appearance (e.g., smoky quartz, gypsum). Opaque minerals do not allow light to travel through (e.g., gold, copper, pyrite).
- Magnetism. This describes the magnetic property of a mineral. Magnetite is an example of a strongly magnetic (strongly attracted to magnets) mineral. There are also moderately and weakly magnetic minerals such as chromite, ilmenite, and columbite. A lodestone is a type of magnetized magnetite that can magnetically attract other materials.
- Effervescence. This describes a mineral’s reaction to a strong acid such as HCl (hydrochloric acid). This is due to the chemical reaction between CaCO3 and HCl in carbonate minerals and rocks. Highly effervescent minerals like calcite exhibit intense “fizzing” or “bubbling” when exposed to HCl. Some minerals are weakly effervescent and only show light “fizzing,” such as rhodochrosite and azurite.
- Odor and Taste. You may have heard that some geologists lick rocks. While that may seem a bit wacky and weird, geologists indeed lick and even smell rocks to identify them. Halite, more popularly known as “rock salt,” is a mineral that gives off a salty taste. Other examples are borax which gives off a sweet taste; epsomite which tastes bitter; and chalcanthite, which is sweet but slightly poisonous. Their “rotten egg” smell can identify sulfur and pyrite.
To this date, thousands of different minerals have been identified and named, and the list grows every year! However, only a few of these minerals are abundant on the Earth’s crust, called rock-forming minerals.
Out of all the elements, only 8 make up most rock-forming minerals.
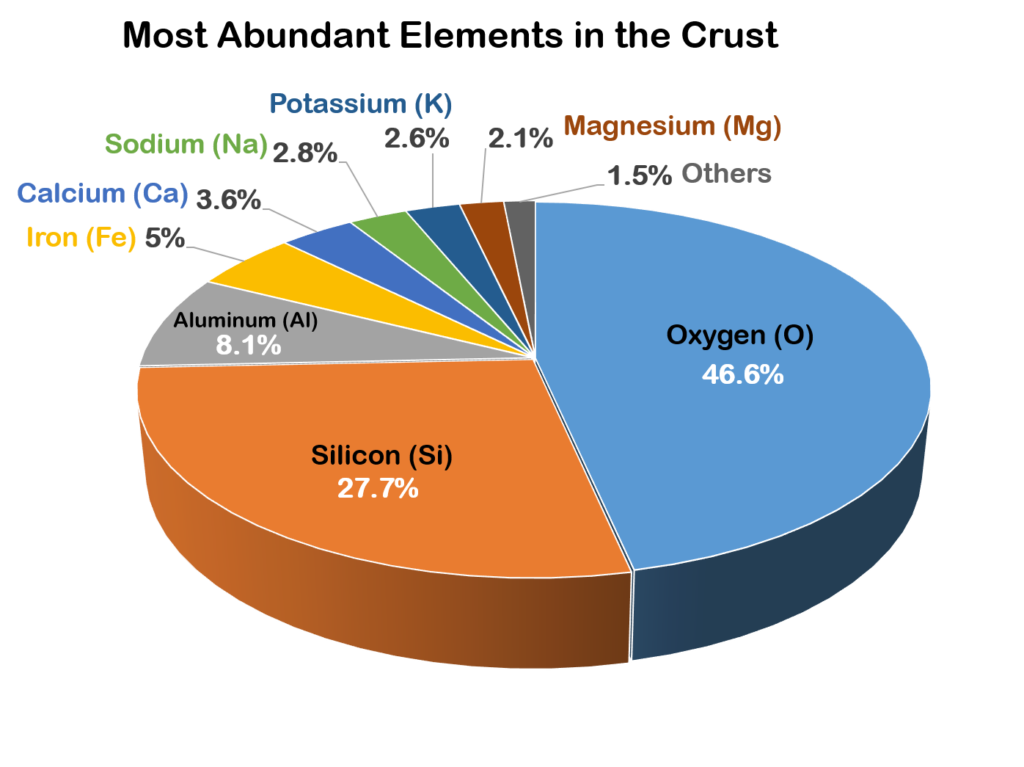
Since oxygen and silicon are the two most abundant blocks, the most common mineral group silicates uses these elements as their “building blocks.”
The other less abundant mineral group, called non-silicates, is further subdivided into groups based on their dominant anion or anionic group. Under non-silicates are the mineral groups native elements, carbonates, oxides, sulfates, phosphates, and others.
Module 3 Practice Test
4. Rocks
Rocks are naturally-occurring aggregates of minerals and mineraloids. There are three main classifications of rocks based on how they were formed.
a. Igneous Rocks
Igneous rocks (from the Latin word ignis meaning “fire”) are formed when molten material cools and solidifies.
When igneous rocks form below the surface of the Earth, they are called intrusive igneous rocks or plutonic rocks. When they form on the surface, they are called extrusive igneous or volcanic rocks.
Intrusive and extrusive rocks can generally be distinguished using the size of their mineral grains. Intrusive rocks have bigger or coarser grain crystals, while extrusive rocks have smaller or finer crystals. This is because higher temperatures beneath the Earth’s surface slow down the cooling rate of minerals, giving more time for larger crystals to form.
The composition of igneous rocks largely depends on what type of magma or lava they form from. The composition of magma depends on the amount of silica (SiO2), which affects its viscosity, and temperature.

Komatiite is a very rare type of extrusive igneous rock (although it can also be classified as intrusive depending on its origin) that forms when extremely hot lava cools rapidly and is common during the Archean eon. However, current surface conditions do not allow komatiite to form anymore.
Other more common types of extrusive rocks are obsidian (formed when lava rapidly cools; also known as volcanic glass), bombs (rounded solidified lava fragments), blocks (angular solidified lava fragments), and volcanic ash.
b. Sedimentary Rocks
Sedimentary rocks are formed from loose material called sediments that have been eroded in weathering and then buried and compacted in a process called diagenesis.
The sediments that make up sedimentary rocks can come from pre-existing rocks and materials or the remains of living things. Because of this, there are two main classifications of sedimentary rocks:
i. Clastic Sedimentary Rocks
Sediments come from pre-existing rocks. Clastic sedimentary rocks are classified based on the characteristics of their clasts, such as size, angularity/roundedness, and sorting.

ii. Non-clastic Sedimentary Rocks
Chemical sedimentary rocks are formed when water evaporates, leaving behind dissolved minerals. Common examples include halite or rock salt, gypsum, flint, chert, travertine, umber, and limestone rocks.

Another type, called biochemical or organic sedimentary rocks, is composed of the remains of living things (shells, bones, plant fragments, etc.). Common examples include some fossiliferous limestone (contains fossils), chalk (composed of very tiny marine organisms called coccolithophores and foraminifera), coquina (composed of >2 mm shell fragments and grains), and coal (altered rock from remains of plant life).
c. Metamorphic Rocks
When a rock is subjected to certain chemical (addition or removal of chemicals) or physical (change in temperature or pressure) processes that alter its chemical composition, mineralogy, and/or texture, a metamorphic rock is formed.
The original rock or “parent rock” that was altered is called a protolith. Metamorphic rocks are divided into two types based on their texture.

The first type is characterized by the appearance of a planar arrangement of mineral grains called foliation or foliated rocks.
Foliation in rock is the result of deformation; the more foliated a rock, the higher the grade of metamorphism.

Nonfoliated rocks usually develop in environments where deformation is minimal and other factors, such as chemically-active fluids, play a larger part in altering the rock. Some common examples of non-foliated rocks are marble, hornfels, quartzite, and metapelite.
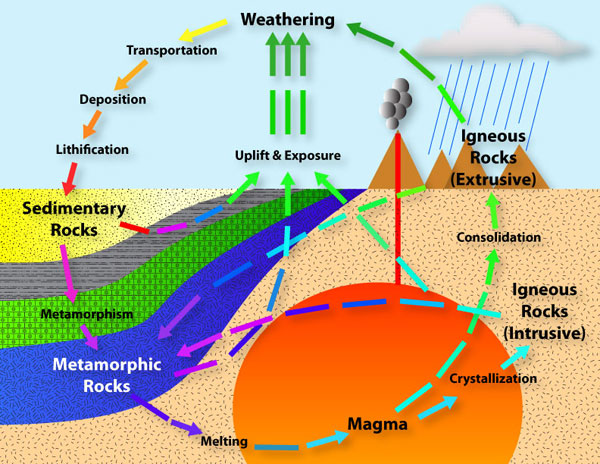
Rocks continually undergo changes that alter their characteristics, ultimately changing them into different rocks.
Here’s a good illustration of the rock cycle which shows what kind of processes can happen to a rock over spans of geological time:
Module 4 Practice Test
5. Depositional Environments, Landforms, and Waterforms
Depositional environments combine chemical, physical, and biological aspects that dictate what type of sediments, rock types, and landforms are deposited or formed.
An essential part of depositional environments, erosion is a geological process in which earth materials are weathered and transported. Erosional agents such as water, wind, ice, or animals and humans are responsible for transporting these materials.
These earth materials are then “added” to an environment or landform in a process called deposition. There are many types of depositional environments, but they can be classified into three main types:
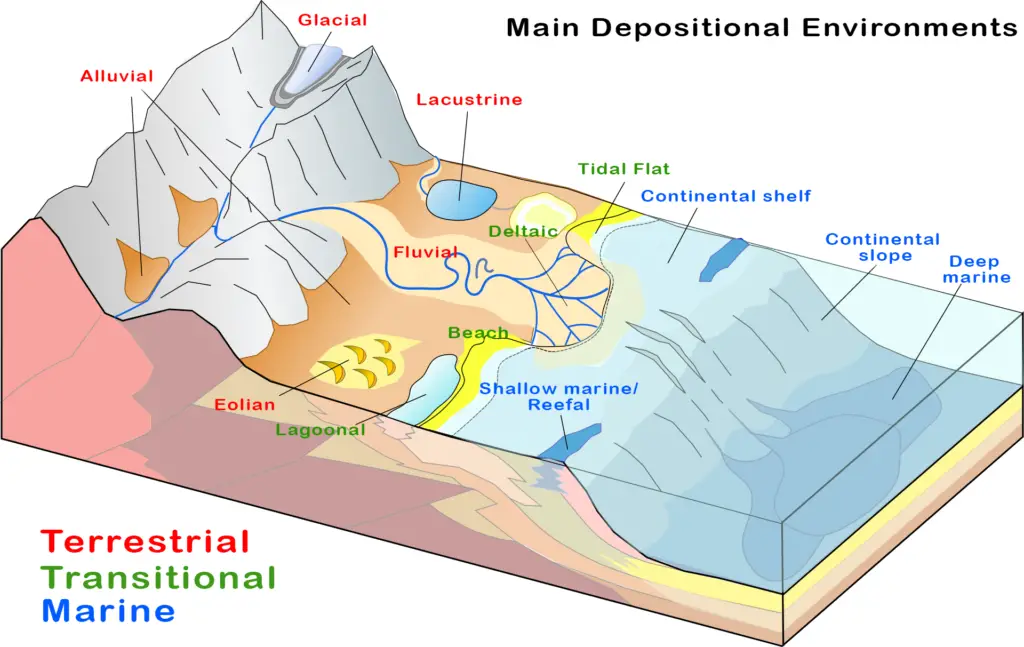
a. Terrestrial Environments
Land and water forms in this environment can be found on land and usually involve freshwater. Here are some depositional environments that fall under this category:
- Fluvial: rivers and streams
- Eolian: deserts and arid environments
- Alluvial: mountainous environments
- Glacial: ice caps and glaciers
- Lacustrine: lakes
Associated landforms and waterforms:
- Mountains – These are elevated (more than 2,000 ft) areas of land, usually resulting from tectonic forces. Hills are similar to mountains but with lesser steepness (below 2,000 ft).
- Plains – These are relatively flat expanses of land that lie above sea level. Plains can occur between two mountains as a valley. A plateau is a plain that is relatively elevated than the surrounding land.
- Desserts – These areas receive little rainfall and have high evaporation rates. Despite this, the most dominant agent of erosion in these areas is running water, followed by wind.
- Glacial environments – These are areas where the most dominant erosional agent is ice. Glaciers are large masses of moving ice over land. Ice sheets are also large masses covering an extensive land area (more than 50,000 km2).
- Rivers – Long bodies of water that originate from high elevation (such as mountains or hills) and flow down to lower elevation (such as plains, mountain slopes, etc.). Rivers are usually supplied with water from rainfall, melted ice, or natural springs from underground in areas called drainage basins.
b. Transitional Environments
Transitional environments represent the interface between land and sea. It is here where freshwater meets with seawater.
Here are some depositional environments that fall under transitional environments:
- Beach: where land meets the sea in shallow waters
- Deltaic: where the river flows into the sea; freshwater mixes with seawater
- Tidal flat: low-lying areas affected by tides
- Lagoonal: a small body of water closed off from a larger body of water (the ocean)
Associated landforms and waterforms:
- Deltas are areas at the end of the mouth of a river where freshwater mixes with seawater.
- Wetlands are areas near rivers or coastlines where soils are saturated or submerged in water. Swamps are wetlands where trees dominate plant life. Marshes are wetlands where moss and soft-stemmed vegetation are most prominent.
c. Marine Environments
These environments can be found in the open waters, from the shallow depths to the deepest portions of the ocean.
Here are examples of marine environments:
- Shallow marine/reefal: a region where sunlight penetrates the water; high energy environment and teeming with life
- Continental shelf: extensions of continental crust submerged by water
- Continental slope: steep slope between the shallow continental shelf and the deep ocean basin
- Deep marine: a region where sunlight does not reach; low energy environment
Associated landforms and waterforms:
- Oceans – These are large bodies of water that surround continents. Seas are smaller bodies of saltwater enclosed or partially enclosed by land and are connected to the ocean.
- Atolls are rings or partial rings of coral that usually form around a volcanic island or volcano that has receded or been eroded.
- Guyots are elevated platforms with flat tops formed by volcanic activity near the ocean floor. These can be massive and reach heights of up to more than 600 m. They are also known as seamounts.
Module 5 Practice Test
6. Basics of Stratigraphy
Stratigraphy is a branch of geology that studies rock layers, beds, or strata (singular: stratum). It is a discipline that correlates rocks and time, helping us understand how, why, and when a certain configuration of strata came to be.
Principles of Stratigraphy
In the 17th century, a Catholic priest named Nicolaus Steno formulated the guiding principles of stratigraphy.
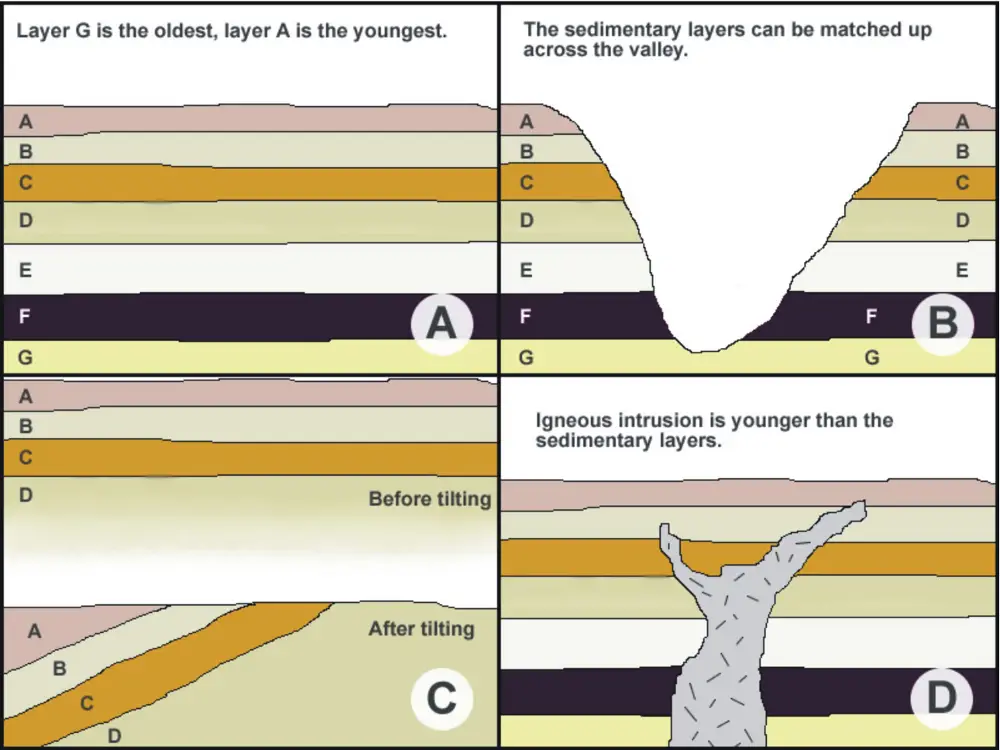
A. Law of Superposition. If the sequence is undisturbed, the layers on the bottom are the oldest, while the layers above are younger.
B. Law of Lateral Continuity. Each stratum extends laterally until it encounters a barrier or obstacle.
C. Law of Original Horizontality. Strata are deposited horizontally.
D. Law of Cross-cutting Relationships. If a geologic body (like an intrusion) or discontinuity (like a fault) cuts across strata, it must be younger than the strata it cuts. An intrusion is an igneous rock body that forms when magma cuts through sedimentary layers and solidifies before it reaches the surface.
In the 19th century, an English geologist named William Smith applied these principles and produced the first geological map of Britain. Since then, he has been regarded as the Father of English Geology. He also introduced another essential principle of stratigraphy:
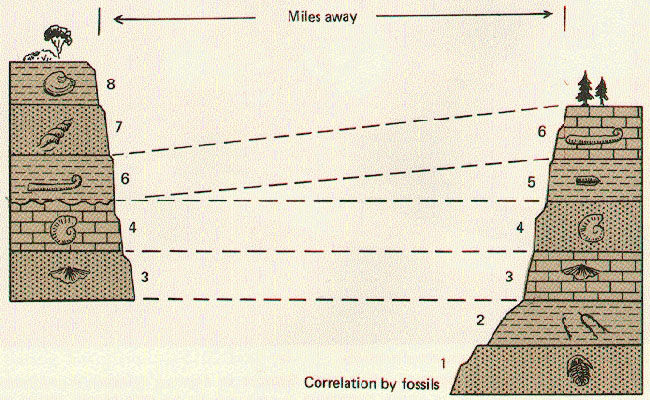
E. Principle of Faunal Succession. Sedimentary strata may contain fossils of plants and animals in a definite and invariable sequence. Thus, the age of a stratum and another stratum in a different location can be correlated if they share the same fossil assemblage.
Sometimes, different processes can lead to a gap in a rock sequence called an unconformity. A hiatus is the “missing time” represented by the unconformity in a rock sequence. There are three types of unconformities:
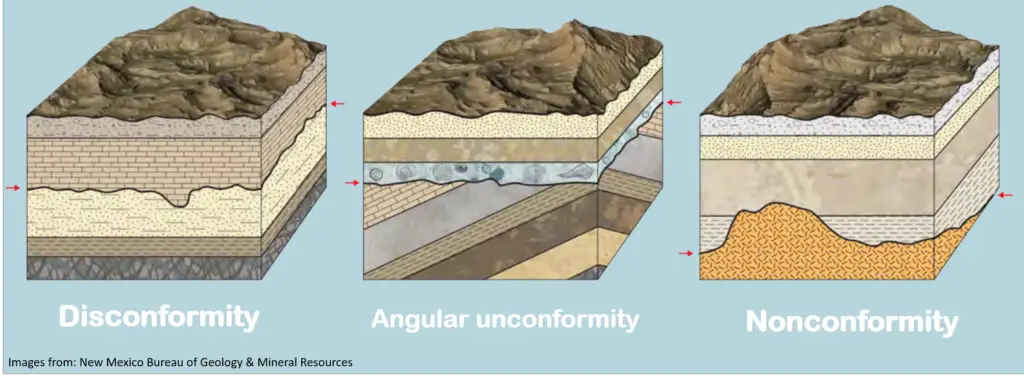
- Disconformity. This type of unconformity is present when there is a missing stratum or strata in the sequence, usually due to a period of non-deposition or erosion.
- Nonconformity. This occurs when sedimentary strata are deposited on top of igneous or metamorphic rock bodies.
- Angular unconformity. When strata are disturbed by forces that cause folding, tilting, and/or faulting, they no longer appear horizontal. The surface is then exposed to erosion, and another set of sedimentary strata is soon deposited on top of the disturbed sequence.
These principles and unconformities can be used to identify the age of strata in relation to other strata in a method called relative dating. However, this method cannot identify a stratum’s specific or absolute age.
Absolute Dating
Determining the absolute age of a layer requires certain techniques collectively known as absolute dating.
One of the best ways to date the numerical age of a rock is to use an absolute dating method called radioisotopic dating or radiometric dating.
As you’ve learned in chemistry, isotopes are atoms of an element with different numbers of neutrons and, thus, different atomic masses. Radioactive isotopes are unstable (parent isotopes) and lose subatomic particles or energy over time in a process called radioactive decay.
Eventually, the parent isotope’s configuration reaches a more stable configuration and becomes a daughter isotope. The half-life of a radioactive isotope refers to the time it takes for half of the atoms in a substance to decay.
Depending on the rock type, different radiometric dating methods can be used. Here are some of the most common radiometric dating methods:
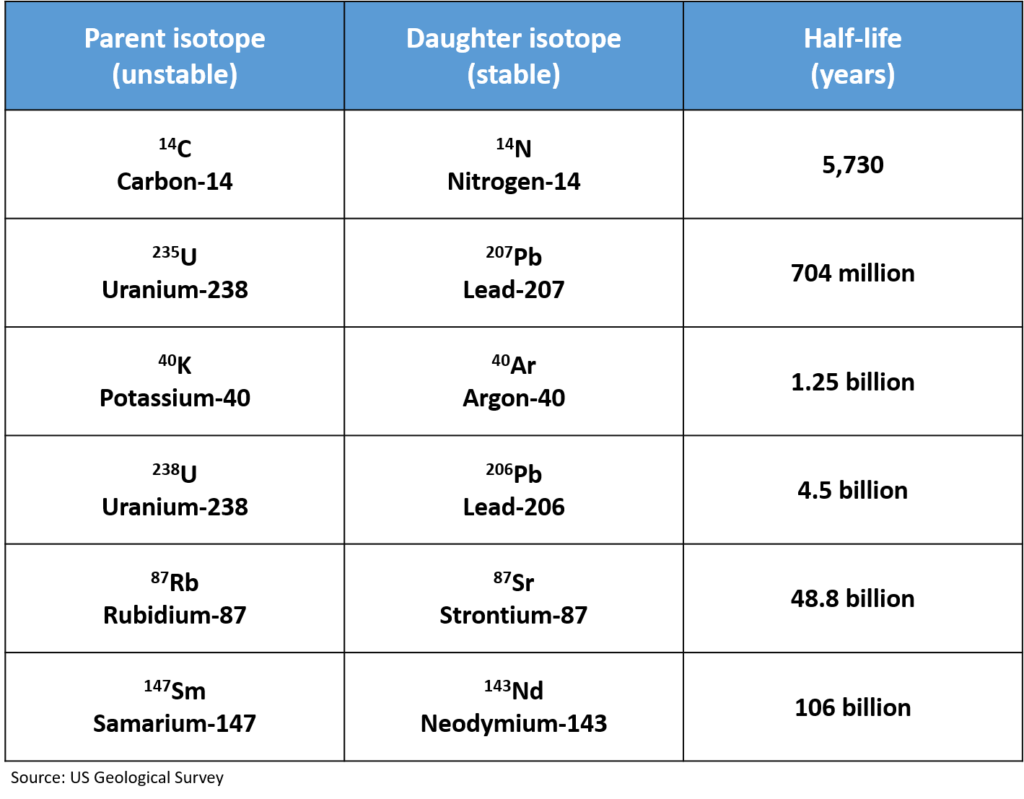
One important thing to note in the table above is that even though 14C – 14N Dating is a common dating technique, it does not date the age of the sediments of the rocks. Rather, it is commonly used to date fossils (which contain C) in a rock.
Fossils are essential to stratigraphy and serve as a doorway to know more about prehistoric life. We shall discuss more about fossils in the next portion.
Module 6 Practice Test
7. Fossils and the Geologic Time Scale
Fossils are the remains of life that are preserved within sediments and sedimentary rocks.
Paleontology is the study of fossils linking concepts of geology and biology to understand prehistoric life over geologic time.
When an animal or plant dies, decomposition, scavengers, and other natural factors usually remove the soft parts of the organism. So to produce a fossil, two conditions must be observed:
- The organism must possess hard parts (bones, teeth, etc.), and
- Rapid burial of the remains increases the chance of preservation.
Different Ways a Fossil Can Be Preserved
a. Permineralization

This occurs when pores and open spaces in tissue (such as bone and wood) are filled with minerals precipitated from mineral-rich solutions such as groundwater.
An example of permineralization at work is when silica precipitates inside the wood’s pores, creating petrified wood. The image above is an example of a permineralized dinosaur vertebra.
b. Molds and Cast

When organisms buried in sediment dissolve or decay away, it leaves behind a hollow space called mold in the organism’s shape. If minerals eventually fill in this hollow space, a cast is made.
c. Amber

Organisms in amber are exceptionally preserved well, often still containing their soft parts. These organisms are preserved when they fall into a viscous tree sap which hardens into amber.
d. Carbonization

Soft-bodied organisms and delicate plant parts can be conserved via carbonization. This happens when these organisms are buried in sediment and eventually dissolve, leaving behind a thin layer of carbon outlining the organism’s shape.
e. Freezing

Organisms can also be exceptionally preserved when they are encased in ice. The image above is of Lyuba, a baby mammoth found frozen in ice in Siberia.
f. Trace Fossils

A fossil doesn’t only pertain to the actual organism. A fossil can be preserved records of its activities such as tracks, burrows, coprolites (fossilized poop), and gastroliths (stomach stones).
Trace fossils can tell a lot about how an organism lived–how it moved, what it ate, and other types of behavior.
The Geologic Time Scale
The geologic time scale (GTS) is a tool geologists use to classify and date rocks and fossils. Instead of numerical ages, time is divided into eons, eras, periods, epochs, and ages (in descending order of duration).
An international body called the International Commission on Stratigraphy (ICS) maintains the GTS. It aims to create unified terminologies for geologists worldwide to use in stratigraphy.
Boundaries of time units often change, depending on new findings and discoveries. You can get the latest version of the GTS here.
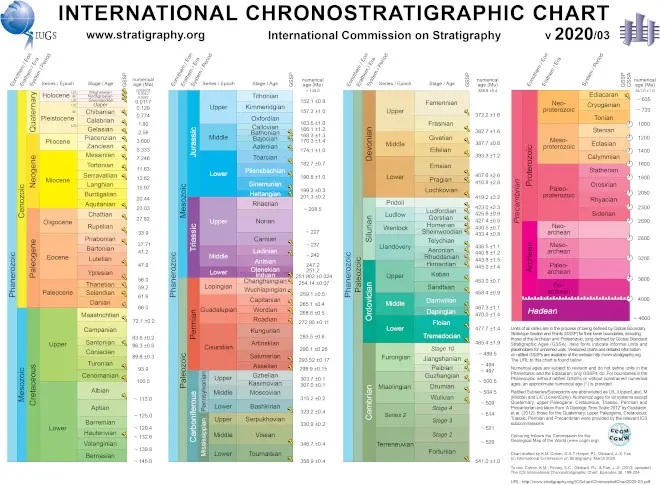
Based on the radiometric dating of the oldest rocks on Earth, the age of Earth is believed to be 4.534 billion years old. Since then, a lot has transpired on our little Earth.
Below is a very, very, very condensed history of the Earth:
From the past…
- Hadean Eon: The formation of the Earth; magma ocean; intense bombardment of space bodies (“Late Heavy Bombardment”)
- Archean Eon: Life begins as prokaryotic bacteria; Blue-green algae start to produce oxygen in the atmosphere
- Proterozoic Eon: Multicellular life emerges
- Cambrian Period: Multicellular life flourishes and diversifies (“Cambrian Explosion”)
- Ordovician Period: “Age of Invertebrates”
- Silurian Period: Emergence of plants on land
- Devonian Period: “Age of Fishes”; Towards the end, true amphibians emerged
- Carboniferous Period: “Age of Amphibians”
- Mississippian: Amphibians diversified; large coal swamps formed
- Pennsylvanian: Emergence of reptiles
- Permian Period: Existence of Pangaea; the largest mass extinction in Earth’s history occurred towards the end (“The Great Paleozoic Extinction”)
- Triassic Period: Dinosaurs emerged; start of the Age of Reptiles; first true mammals (therapsids) emerged as well
- Jurassic Period: Dinosaurs dominated the Earth; the first birds emerged
- Cretaceous Period: first flowering plants emerged (angiosperms); marked the end of the Age of Reptiles with the Cretaceous-Tertiary Extinction (“K-T Extinction”)
- Paleogene Period: start of the Age of Mammals
- Neogene Period: Mammals and birds evolved into modern forms; hominids, the ancestors of humans, appeared towards the end
- Quaternary Period: current period; a cycle of glacial and interglacial periods
…to the present!
Module 7 Practice Test
8. Plate Tectonics
The Continental Drift hypothesis paved the way for the emergence and acceptance of the plate tectonics theory. It was proposed by a German meteorologist and geophysicist named Alfred Wegener.
Wegener hypothesized that long ago, there was a supercontinent that consisted of all landmasses on Earth. He named this supercontinent Pangaea (from the Greek words pan meaning “all” and gaia meaning “land”). He and other supporters of the continental drift hypothesis collected evidence to substantiate their claims.
Evidence of the Continental Drift Hypothesis
Evidence #1. Continental Jigsaw Puzzle
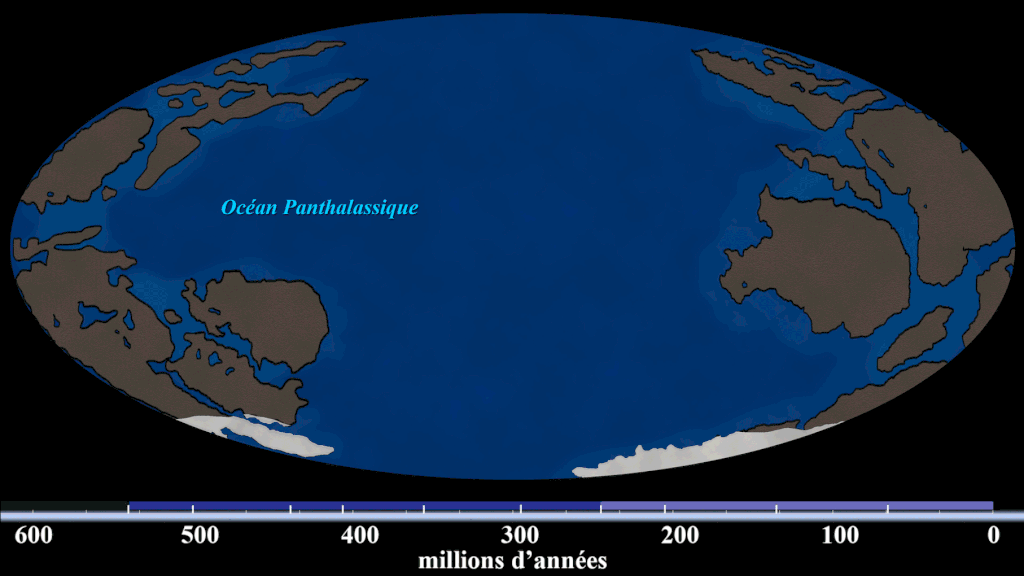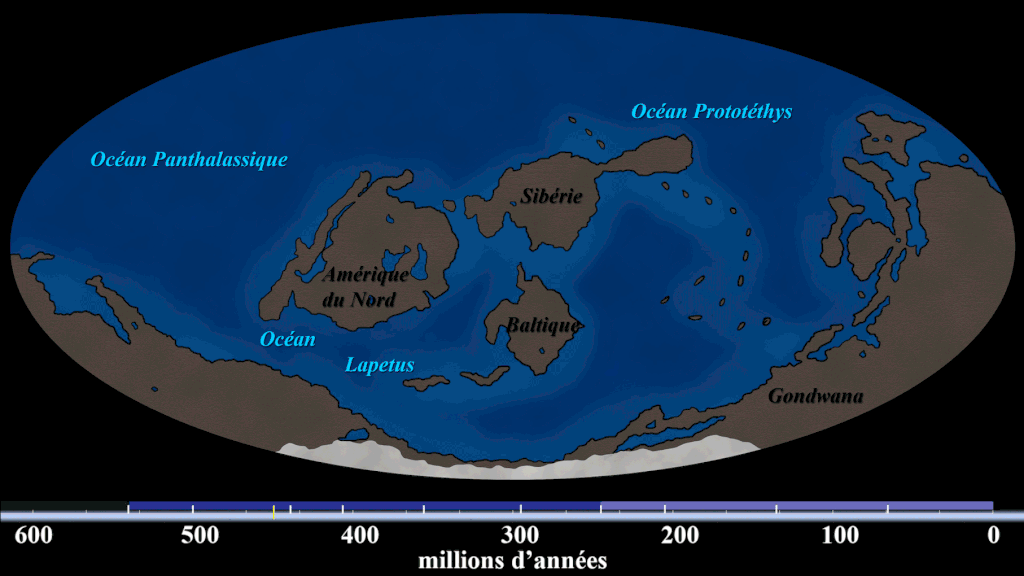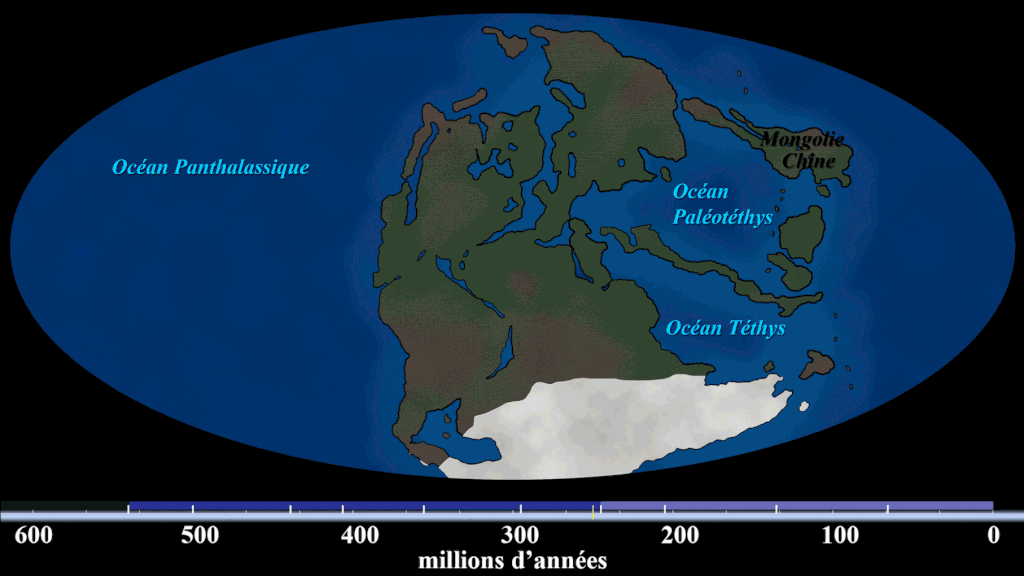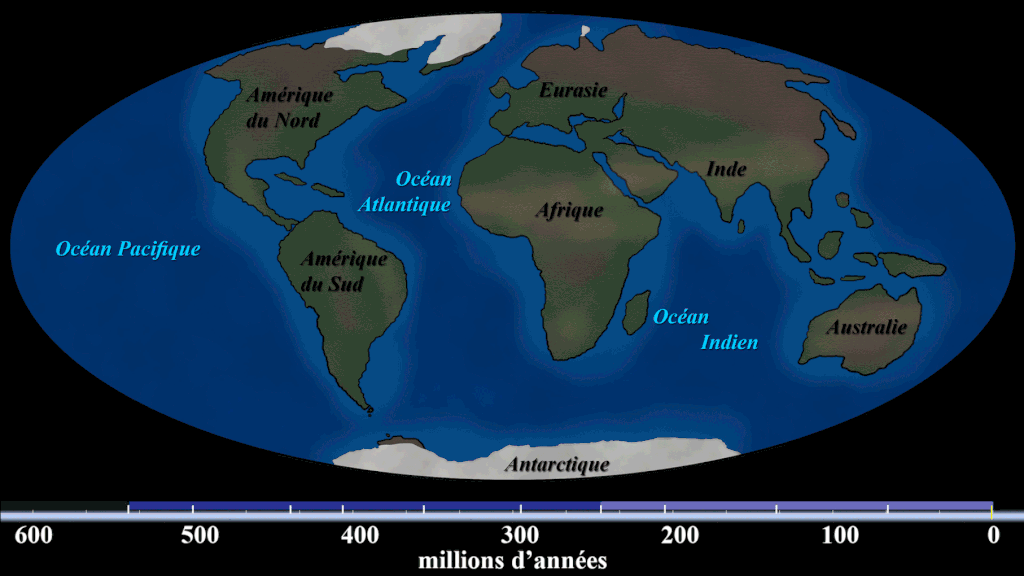
If you take the boundaries of each continent and try to fit them together, you’d get a landmass similar to the configuration of Pangaea. Wegener argued that the excellent fit of the continents was more than a coincidence, citing the almost perfect fit of South America and Africa.
Evidence #2. Fossils
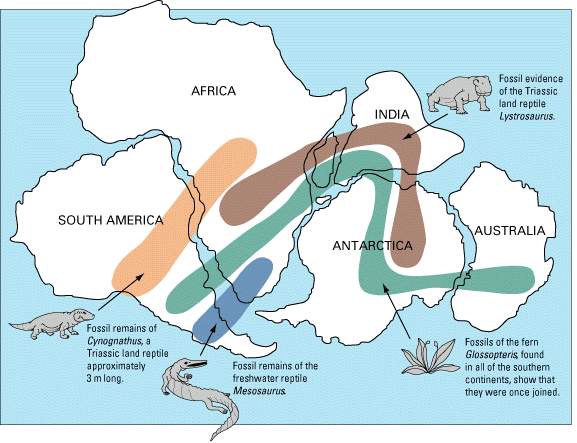
Similar fossil remains of plants and animals were found on continents currently separated by large bodies of water. Paleontologists agreed that these organisms wouldn’t have been able to cross these oceans due to inherent characteristics (e.g., the Mesosaurus lived only in freshwater, the Glossopteris seeds were too heavy to be carried by the wind across great distances, etc.).
Evidence #3. Similar Rock Types and Geologic Features
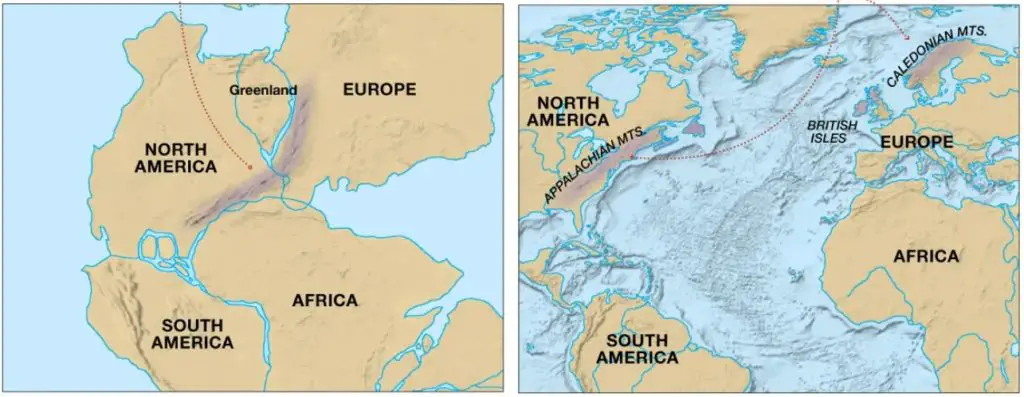
Large mountain belts of similar ages and rock types could be matched with each other across continents. This is the case with the Appalachian Mountains in the eastern margin of North America being similar to the Caledonian Mountains in the western margin of Scandinavia.
Evidence #4. Ancient Climates
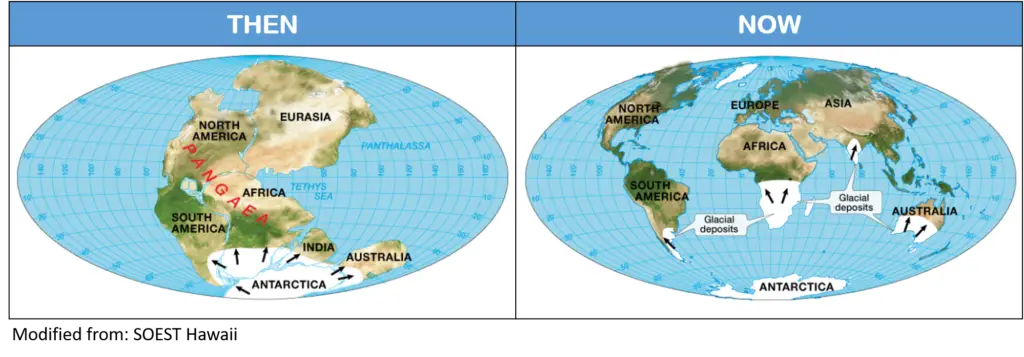
According to Wegener, evidence suggesting there were glaciers before in present-day continents (such as Africa, South America, and Australia) located in the equator supported continental drift. However, the opposition to the hypothesis suggested that this may be due to extreme global cooling.
Wegener asserted this was not the case because evidence showed that large tropical swamps co-existed with the glaciers at the time.
Despite all these pieces of evidence, the scientific community still did not accept the continental drift hypothesis primarily because of one problem: Wegener could not explain how the continents drifted. It wasn’t until after his death would the mystery be solved.
The Development of the Plate Tectonics Theory
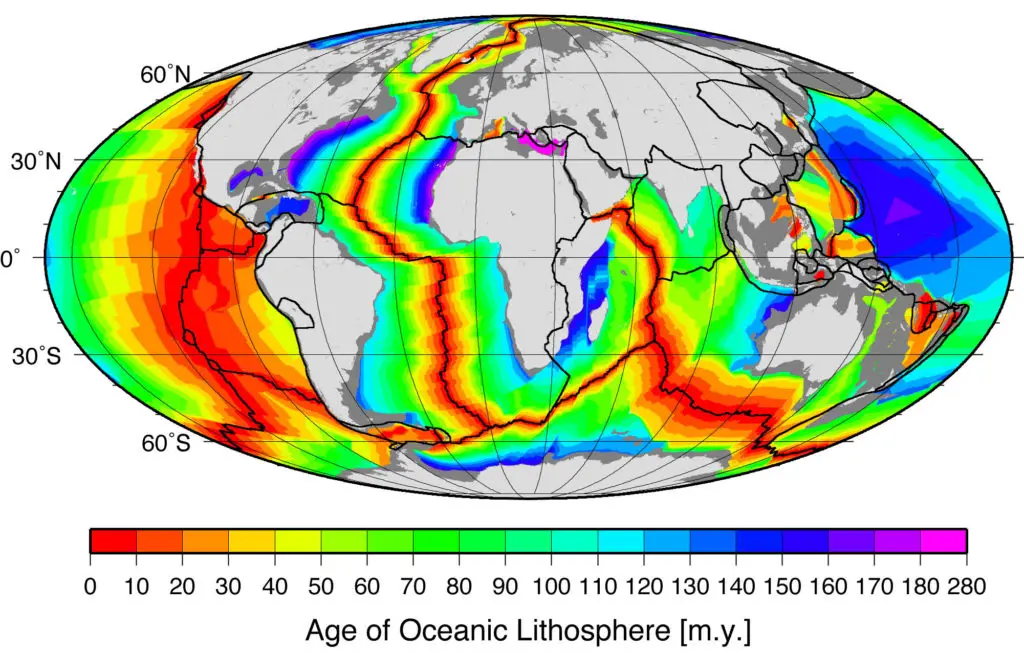
After World War II, extensive ocean exploration led to the discovery of the global oceanic ridge system, which spans the globe, making it the longest mountain range (around 80,000 km long).
New oceanic crust forms in the axis of this ridge system. Because of this, rocks become progressively older and thicker with sediment away from the axis. This phenomenon was termed seafloor spreading by Harry Hess and Robert Dietz.
However, the dredging of the ocean floor showed that the oldest oceanic crust was no more than 180 million years old. If the new oceanic crust was constantly being generated along the ridge, where did the old oceanic crust go? The theory of plate tectonics addresses this question.
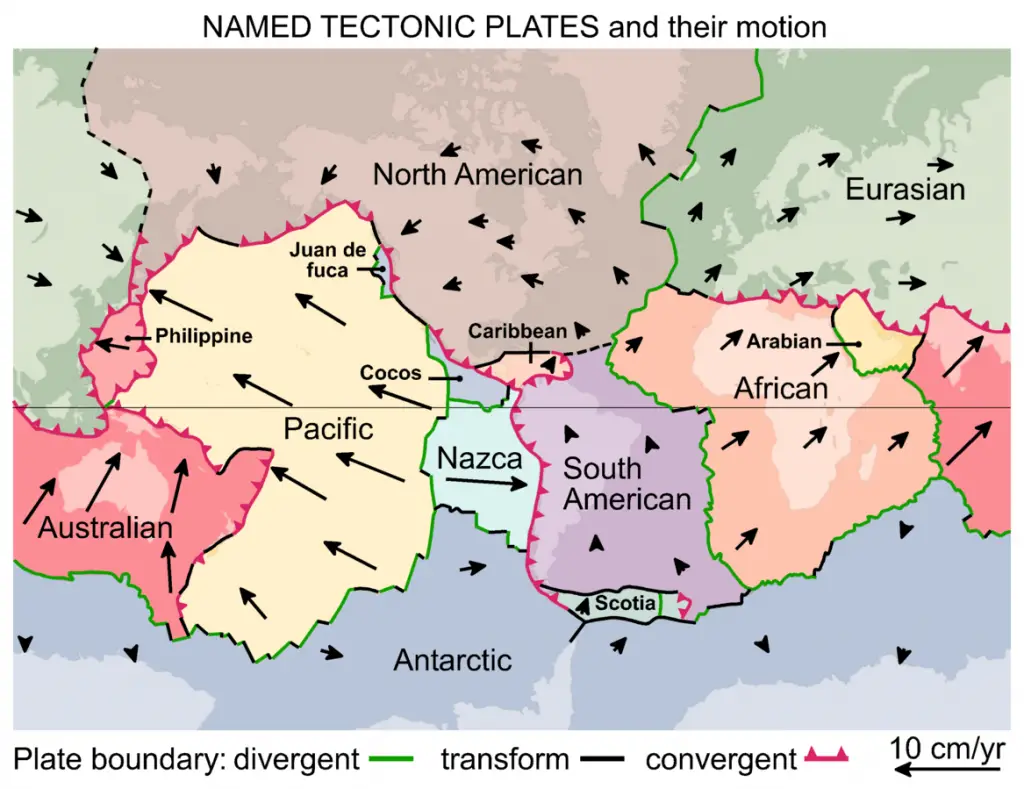
The plate tectonics model states that the lithosphere is broken into rigid slabs called tectonic plates or simply plates. These plates overlie the ductile asthenosphere, allowing them to be in constant motion concerning one another.
Seven major plates cover 94% of the Earth’s surface area:
- African plate
- Antarctic plate
- Eurasian plate
- Indo-Australian plate
- North American plate
- Pacific Plate
- South American plate
There are also minor plates such as the Philippine Sea plate, Juan de Fuca plate, Cocos plate, Nazca plate, Scotia plate, and Arabian plate.
As mentioned earlier, plates are always in constant motion. Because of this, the plates’ margins always interact with one another. The sites where these margins interact are called plate boundaries.
Three Main Types of Plate Boundaries
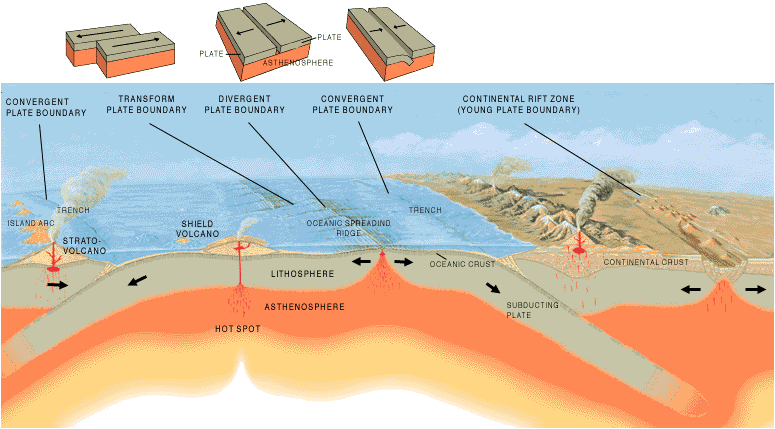
a. Divergent Plate Boundaries (Constructive Margins)
Divergent plate boundaries are formed when two plates move apart relative to each other. These plate boundaries are also called constructive margins because the pulling apart of two plates results in the migration of molten material from the mantle to the surface, generating a new crust. Divergent boundaries can be found both on the ocean floor and inland.
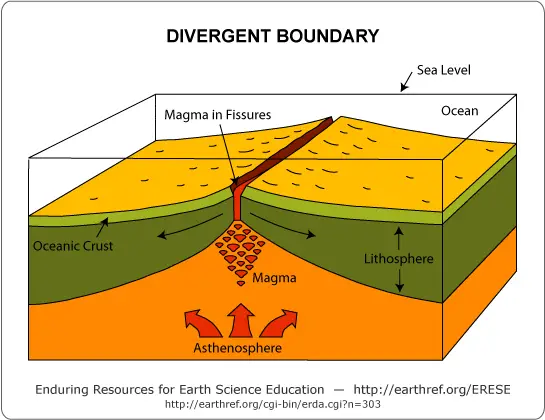
Divergent boundaries on the ocean floor manifest as the oceanic ridge system, which was discussed earlier.
New seafloor is generated via seafloor spreading. As the seafloor gets older, it gets denser and moves toward the edge of the plate. The younger oceanic crust is hot and less dense, while the older oceanic crust is cooler and denser.
The spreading rate varies along the oceanic ridge system, going as slow as 2 cm/year or as fast as 15 cm/year.
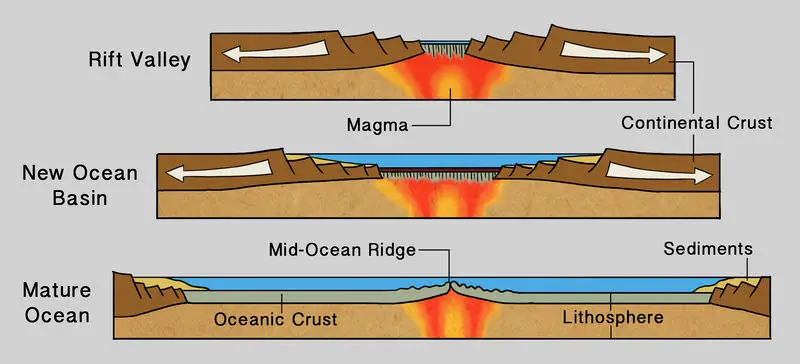
Divergent boundaries within a continent generate an elongated depression called a continental rift. These rifts form by the stretching and thinning out of the lithosphere.
The rift valleys can grow wide enough to split the continent apart, producing large depressions. These depressions would eventually be filled with water, producing new ocean basins.
A good modern-day example of continental rifting at work is the Red Sea in the East African Rift in Eastern Africa.
b. Convergent Boundaries (Destructive Margins)
Convergent boundaries are the sites where plates move towards each other, resulting in a collision or one plate going under the other in a subduction process. They are called destructive margins because the crust is consumed in the process. There are three scenarios in which convergence could occur:
i. Oceanic-Continental Plates Convergence
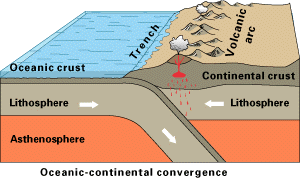
This setup answers our question from earlier: Where does the old oceanic crust go?
In this case, the denser or heavier block subducts or goes underneath the lesser dense block. Because the oceanic crust comprises basalt, it subducts underneath the continental crust of the lighter granitic material in a subduction zone.
In a subduction zone, partial melting is induced in the overlying continental crust, producing volcanic activity called continental volcanic arcs. The subduction of the oceanic crust usually results in large, deep linear depressions on the ocean floor called deep-ocean trenches.
The deepest oceanic trench in the world is the Mariana Trench in the western Pacific Ocean, with a depth of nearly 11,000 km, deeper than the height of Mt. Everest (8,800 km).
ii. Oceanic-Oceanic Plates Convergence
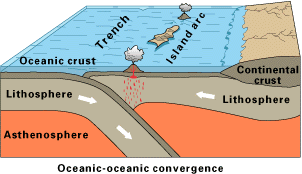
When two oceanic plates collide, the older and denser one subducts.
Much like oceanic-continental convergence, one plate’s subduction generates volcanism, forming a chain of volcanic islands called a volcanic island arc or simply an island arc.
Most island arcs are located in the western portion of the Pacific Ocean, while a few can be found in the Atlantic Ocean.
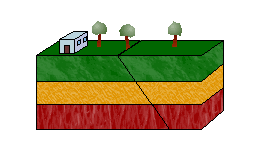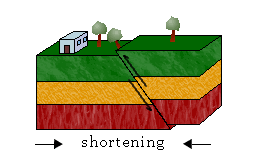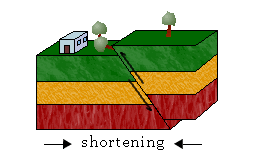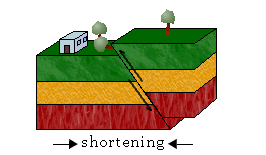
iii. Continental-Continental Plates Convergence
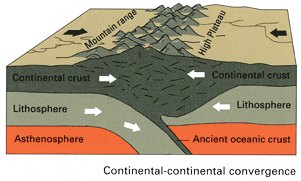
Because continental crust is too thick and buoyant to be subducted, most crustal material is deformed and pushed up instead. This results in the accumulation of sediments and rocks along the margin, forming mountain belts in a process called orogeny.
The most famous example would be the Himalayan mountain range formed from the collision of the Indian and Eurasian plates nearly 50 million years ago.
c. Transform Plate Boundaries (Conservative Margins)
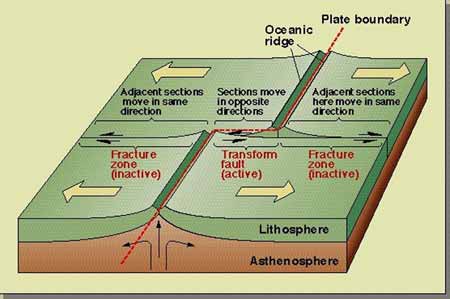
These plate boundaries are characterized by two plates sliding past each other, not destroying or producing new crustal material. They are also called transform faults and are usually found in fracture zones.
Fracture zones are linear breaks on the ocean floor that run perpendicular to oceanic ridges. An active transform fault lies between the two offset oceanic ridges, while the areas beyond the ridge zones are inactive zones.
Below is a diagram showing how different plate boundary types interact with one another:
Module 8 Practice Test
9. Earthquakes
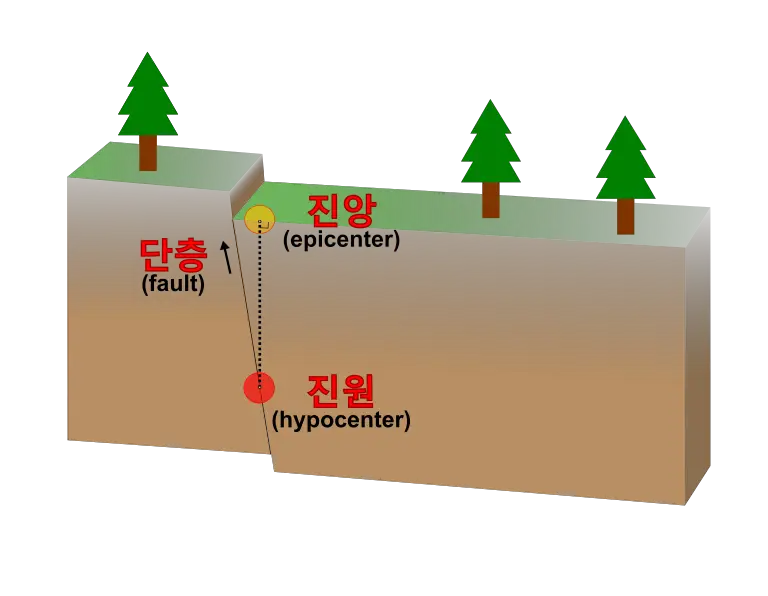
Earthquakes occur when one block of earth slips past another block along surfaces called faults or fault planes and generates ground shaking.
The area under the earth where the slippage originates is called the hypocenter or focus. The epicenter is the point on the Earth’s surface directly above the hypocenter.
Seismic Waves
When slippage happens, the stored energy is released as seismic waves. The seismic waves travel through the earth and cause it to shake. These waves can be classified into two types: body waves and surface waves.
a. Body Waves
Body waves are waves that travel through the interior of the Earth. There are two types of body waves: primary waves (P waves) and secondary waves (S waves).
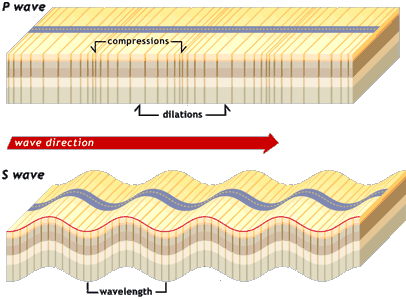
i. Primary Waves
These are the fastest seismic waves and can travel through solid, liquid, and gas. These waves push and pull the rocks in the direction the wave is traveling. They are also called compressional waves because of this behavior.
ii. Secondary Waves
These waves cause the rocks to shake up and down at right angles to the direction of the traveling wave. S waves are slower than P waves and can only travel through solids. Because of this, S waves cannot propagate through the liquid outer core. They are also called shear waves.
b. Surface waves
Surface waves, as the name implies, can only travel on the surface of the Earth. These are the waves that can cause tremendous damage. There are also two types:
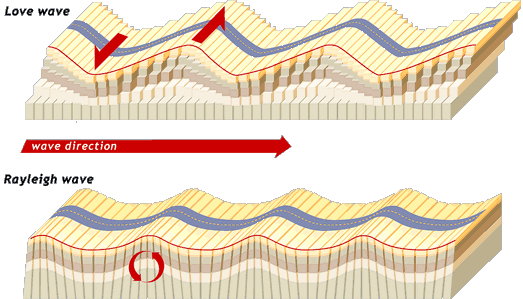
i. Love Waves
These waves are responsible for shaking the ground horizontally and vertically in an S-like pattern.
ii. Rayleigh Waves
These waves move in a rolling motion similar to ocean waves.
Sometimes, before the main earthquake or mainshock, smaller and weaker quakes called foreshocks occur. The mainshock is the largest quake in the sequence.
Weaker and shorter quakes, called aftershocks, usually occur afterward. These quakes may or may not be felt, depending on the size of the mainshock, and can even occur for days, weeks, or even months.
Thousands of earthquakes occur daily worldwide, but most are too small to be felt by people or cause damage.
Seismology
Seismology is the study of earthquakes. Seismologists use seismographs or seismometers to record earthquakes; these tools are sensitive to ground shaking.
To describe and classify earthquakes, the intensity and magnitude are determined.
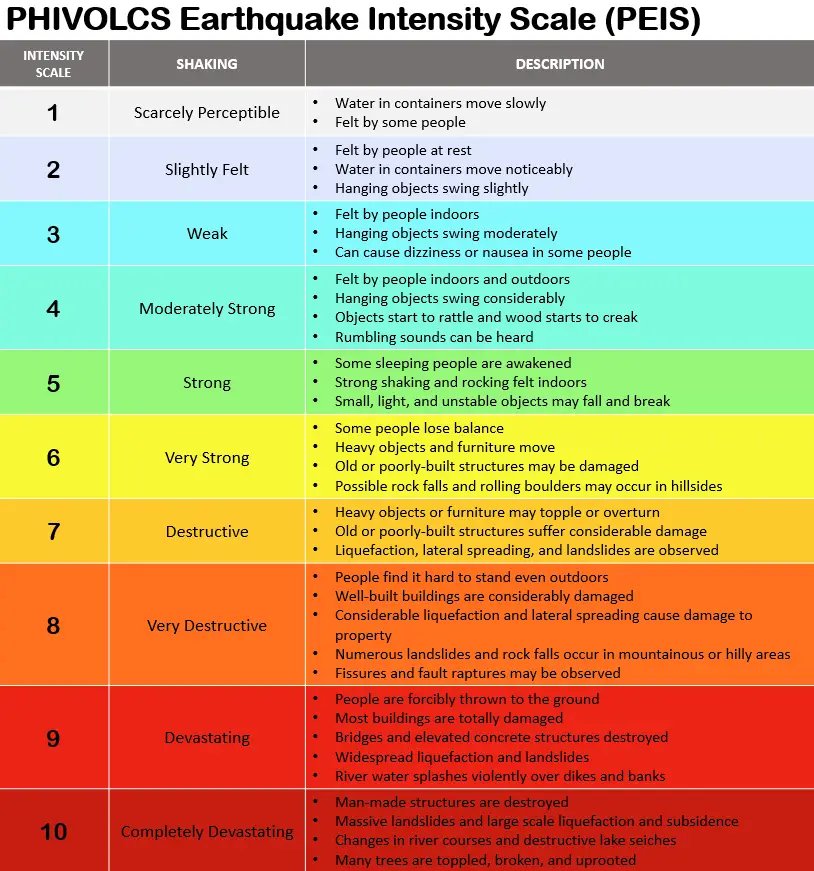
The intensity refers to the qualitative measurement of ground shaking at a particular location, depending on the damage to property, life, and nature.
Different intensity scales are used in different countries. The Modified Mercalli Intensity Scale is used in countries like the United States. However, the PHIVOLCS Earthquake Intensity Scale (PEIS) is used in the Philippines. This scale was developed by the Philippine Institute of Volcanology and Seismology (PHIVOLCS) as a response to the devastating 1990 Luzon Earthquake.
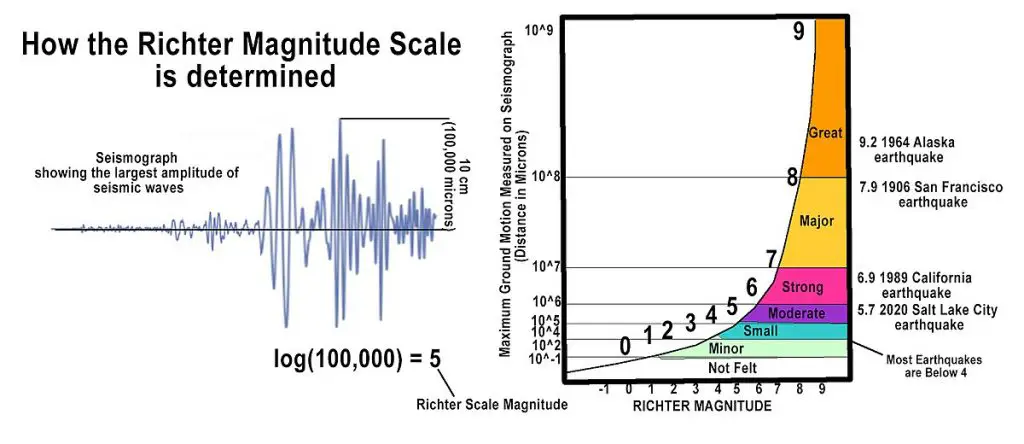
The magnitude refers to the quantitative measurement of energy released at the earthquake’s source.
Before, the Richter Scale was the most commonly used scale for measuring the magnitude, which measures the amplitude of the largest seismic wave on a seismogram. Now, seismologists use the Moment Magnitude (Mw) Scale, which measures the total energy an earthquake releases. The Moment Magnitude scale is more effective in measuring stronger earthquakes (Mw 5 and above) than the Richter scale.
Faults
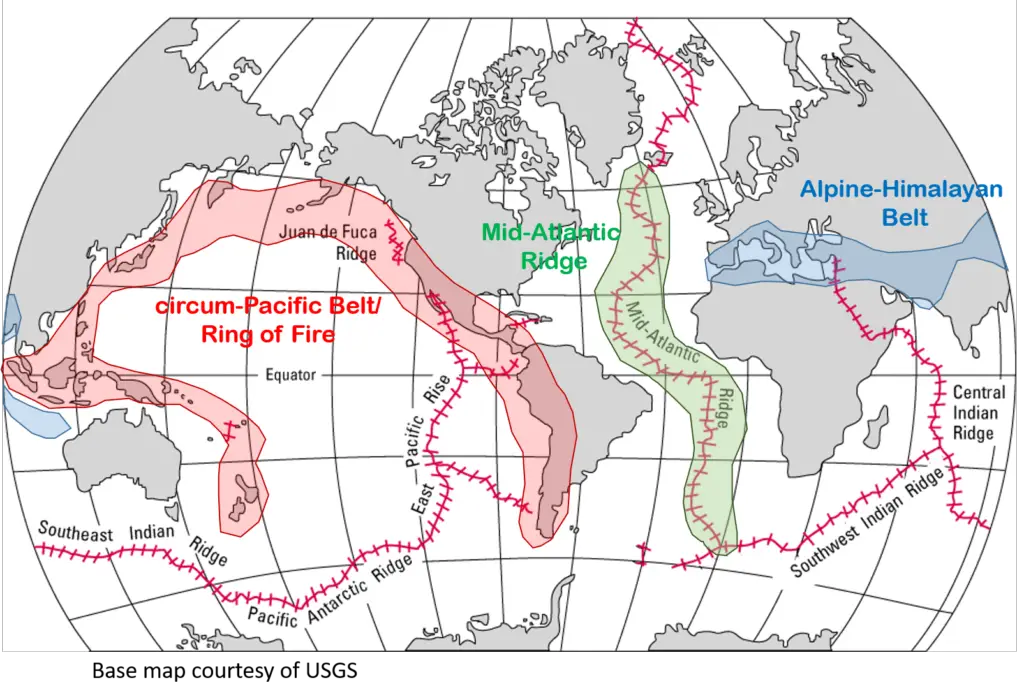
Where do earthquakes come from? Fortunately for some (and unfortunately for others), nearly 81% of earthquakes occur in a tectonically-active region called the circum-Pacific Belt (known as the Ring of Fire).
The next most tectonically-active seismic belt is the Alpine-Himalayan Belt, where 17% of the world’s earthquakes occur. The rest of the earthquakes occur along the Mid-Atlantic Ridge in the Atlantic Ocean.
Earthquakes along the seismic belts originate from convergent plate boundaries. The contact between the two interacting plates is called megathrust faults which can produce earthquakes of Mw 9.0 and above.
Natural or man-made events can cause earthquakes.
Earthquakes caused by the eruption of volcanoes are called volcanic earthquakes. Generally, small earthquakes called collapse earthquakes occur when underground caves or mines collapse. The detonation of explosives can also cause earthquakes called explosion earthquakes.
The most common type of earthquake, tectonic earthquakes, are caused by fault movement. There are main types of faults:
a. Normal Faults
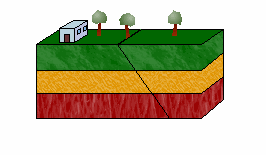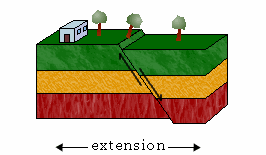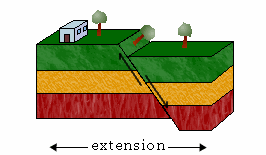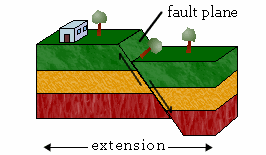
The hanging wall moves down relative to the footwall in a normal fault.
Fun fact: Miners coined the term “hanging wall” to refer to the wall where they would hang their lamps and “footwall” for the surface on which they walked.
Normal faults result from tensional forces that pull the two slabs apart. They are also known as tensional, gravity, or normal-slip faults.
b. Reverse Faults
In a reverse fault, the hanging wall moves up relative to the footwall. This type of fault results from compressional forces pushing the two slabs together, shoving the hanging wall above the underlying block. These are also known as thrust, compression, or reverse-slip faults.
c. Strike-Slip Faults
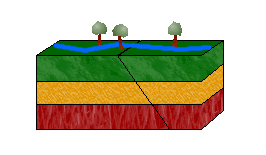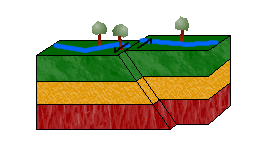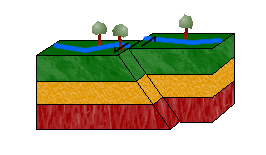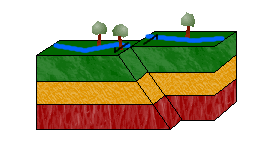
In a strike-slip fault, blocks move horizontally to one another due to shearing forces.
Left-lateral strike-slip faults (or sinistral faults) occur when one block moves to the left relative to the other. Right-lateral strike-slip faults (or dextral faults) occur when the block moves to the right.
An easy way to distinguish between the two is to imagine yourself standing on one block, facing the other. If the block on the other side moves to the left, the movement is left-lateral. If it moves to the right, right-lateral.
Can you guess the movement of the strike-slip fault in the animation above? The answer: left-lateral.
d. Oblique-Slip Faults
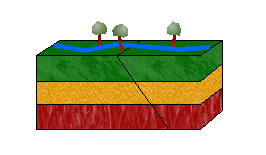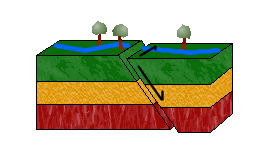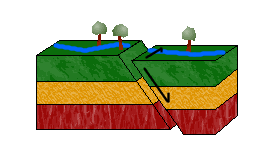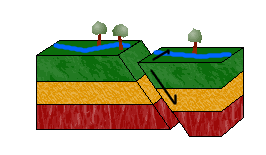
As pictured above, combining shearing and tensional or compressional forces would result in an oblique-slip fault.
Earthquake-related Hazards
As we all know, major earthquakes can devastate living and nonliving things. Depending on the destructive force of the earthquake, it can cause the following events:
a. Landslides and Ground Subsidence
These are caused by ground shaking during an earthquake.
A landslide is a form of mass wasting where large amounts of earth move down a slope under the influence of gravity. They can have devastating effects, especially in heavily populated areas near hillsides or mountain slopes.
Subsidence is the sudden sinking of the Earth’s surface due to the movement of the earth underneath. Liquefaction is similar to subsidence but occurs when sediments are saturated with water. While these events can occur naturally, they are usually aggravated by earthquakes.
b. Flooding and Water-related Hazards
During and after an earthquake, large water pipes underground and dams may be damaged and fail. These can cause flooding in populated areas, resulting in property damage and harm to life.
In enclosed bodies of water such as lakes or reservoirs, waves called seiches may occur. These are oscillating waves that produce major fluctuations in the water level, depending on the strength of the earthquake.
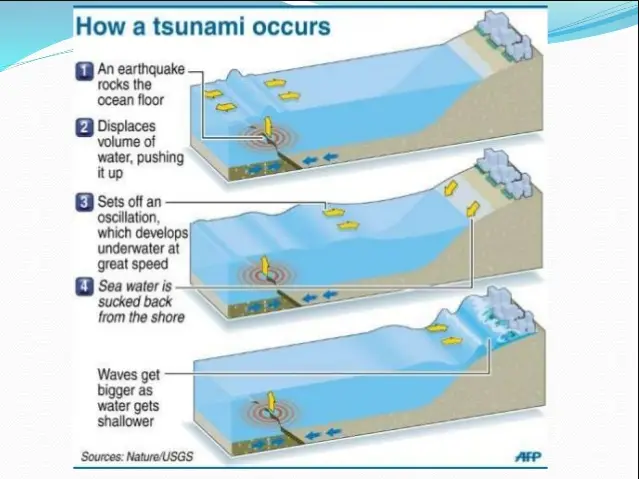
One of the most dangerous effects of earthquakes that originate offshore is tsunamis.
Tsunamis are giant waves that are produced when a fault displaces a large slab of the ocean floor. They are nearly undetectable in the open ocean. Still, once tsunamis reach shallow waters, wave height increases dramatically and can reach up to 30 m, like what happened in the 2004 Indian Ocean Mw 9.1 Megathrust Earthquake in Sumatra, Indonesia.
Related: Midnight Shocker of ’76: Remembering The Deadliest Tsunami In Philippine History
c. Damage to Man-made Structures
Depending on the material used and how they were constructed, structures such as buildings, bridges, roads, dams, and others are susceptible to damage.
Damage can range from cracks in the walls to the destruction of property. Fires can also break out due to severed gas and electrical lines. Coupled with broken water pipelines, even small fires can quickly spread and cause massive damage.
Spillage of hazardous chemicals from factories and chemical containment facilities is also a possible threat, such as the leakage of radioactive water from the Fukushima nuclear power plant during the 2011 Japan Earthquake.
Module 9 Practice Test
10. Volcanoes
Volcanism is a geological process where hot molten rock from underneath the earth reaches the surface through an opening in the ground. A volcano is the most recognizable form of an opening, where molten material flows out onto the surface during a volcanic eruption.
An eruption describes how the molten material was ejected, whether it was violent (explosive eruptions), non-explosive (effusive eruptions), or what caused the eruption (hydrothermal, phreatic, phreatomagmatic, etc.).
The hot, molten material is called magma when it’s underground and lava when it reaches the surface.
How Volcanoes Are Formed
The formation of volcanoes is deeply tied to the theory of plate tectonics. There are three main ways magma can rise to form volcanoes:
a. Convergent Boundaries
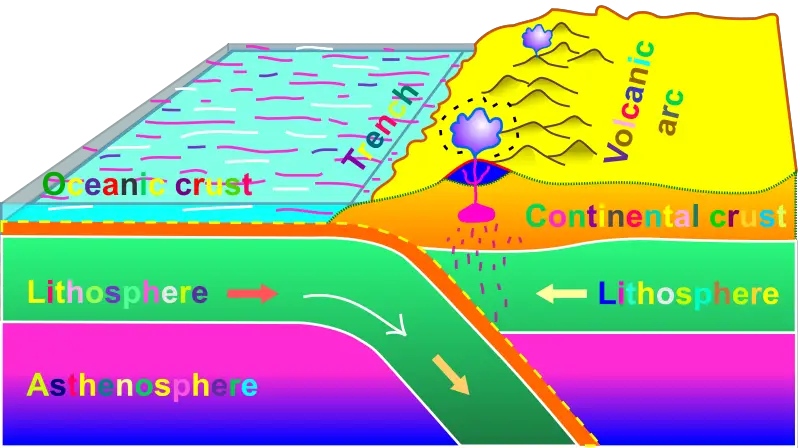
As previously discussed, partial melting occurs in subduction zones, which are responsible for the heating and partially melting of the rocks in the overlying plate.
This is caused by introducing volatiles (seawater, water from minerals, and other fluids) from the oceanic lithosphere, lowering the surrounding rocks’ melting temperature. The molten rock then starts to ascend to the surface in the form of volcanic activity.
b. Divergent Boundaries
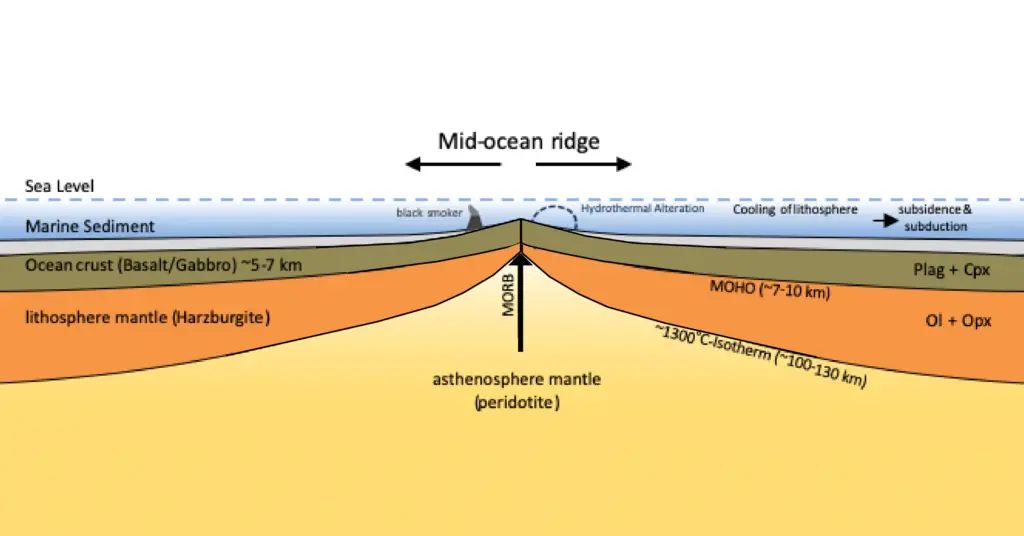
When plates move apart, pressure in the lithosphere reduces, allowing magma in the asthenosphere to rise and induce partial melting of the surrounding rocks. An excellent example of divergent boundary-produced volcanism can be found in the Mid-Atlantic Ridge.
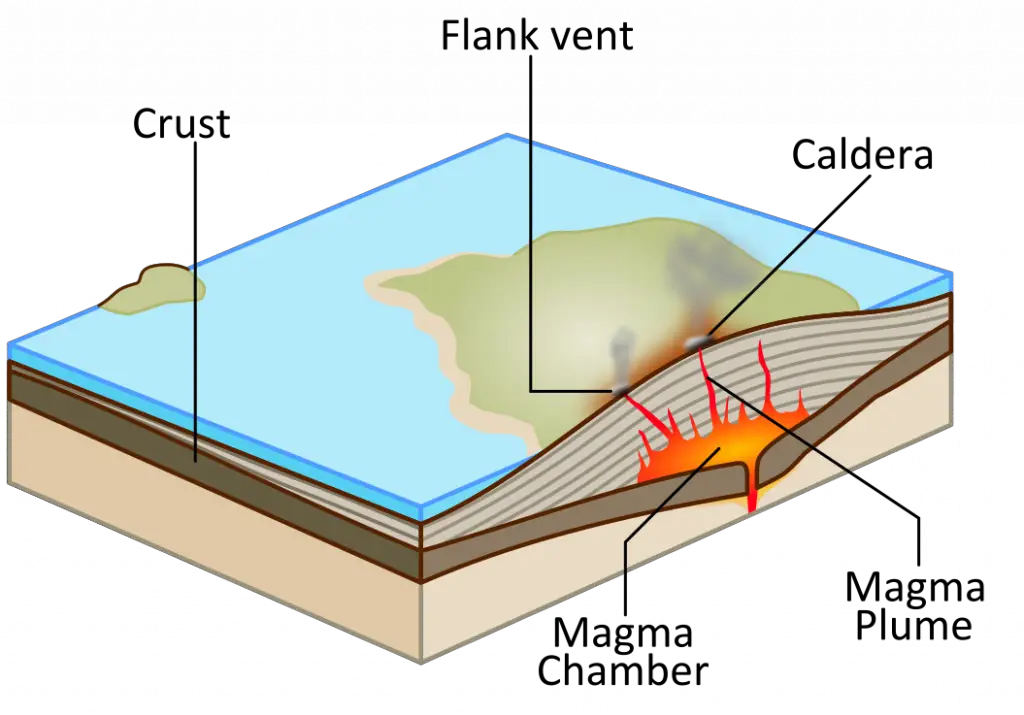
c. Hotspots and Mantle Plumes
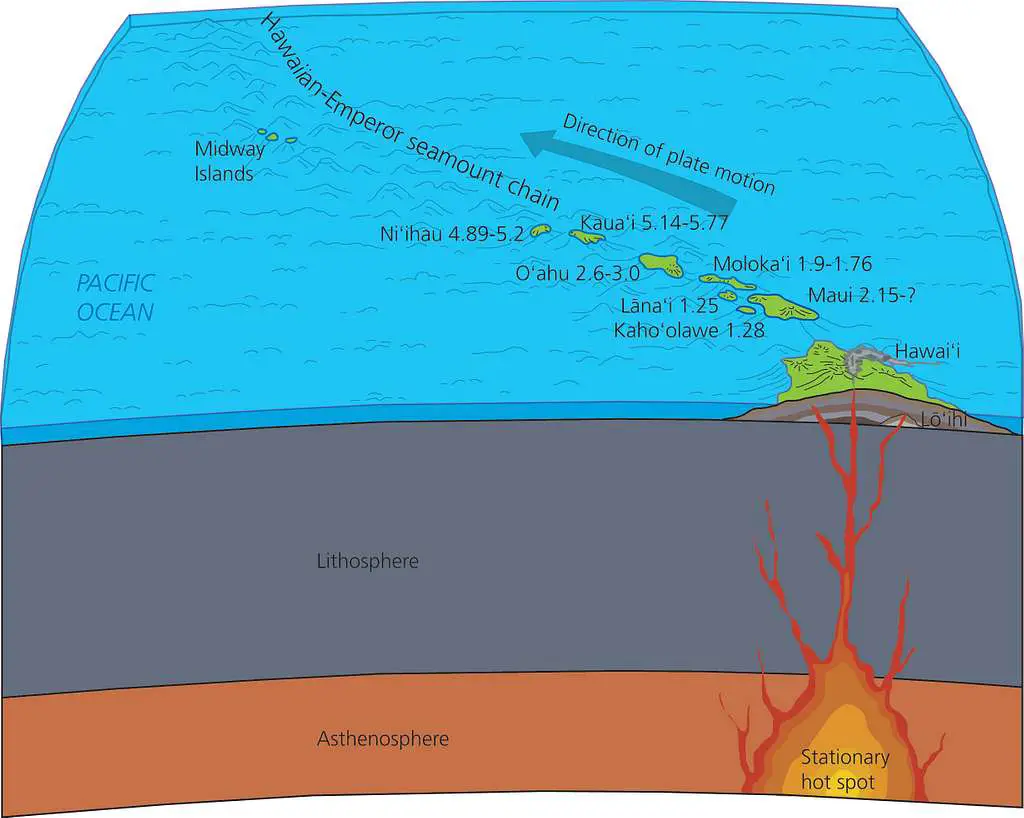
Mantle plumes are areas where the mantle rises towards the surface, originating deep within the mantle. A hotspot is the surface manifestation of a mantle plume.
Unlike the other two ways, volcanism at hotspots does not occur at plate boundaries. As plates move above a hotspot, the increase in temperature induces partial melting and generates hotspot volcanism.
The most famous example of hotspot volcanism is the Hawaiian-Emperor Seamount Chain.
Volcano Morphology
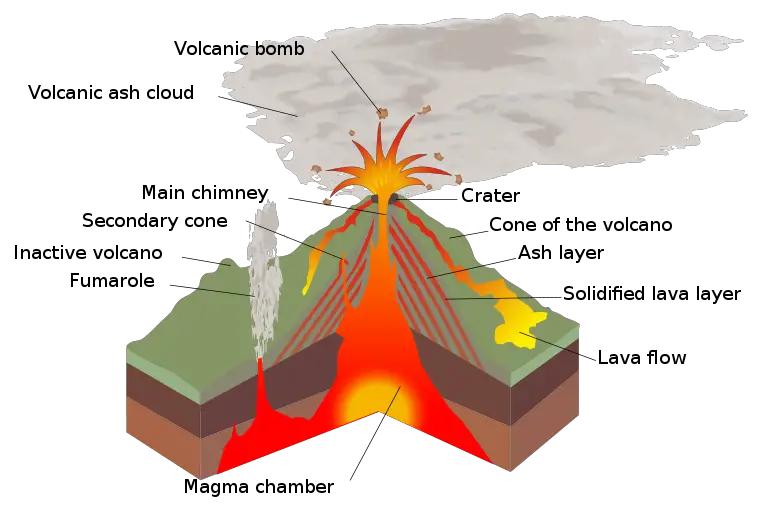
Different types of volcanoes have different shapes and sizes. However, most volcanoes share certain characteristics. Here is the anatomy of a generalized volcano.
- Magma chamber – the reservoir of molten material in the Earth’s crust, replenished with magma from a deeper reservoir in the mantle
- Main vent – the pathway for magma to come to the surface
- Crater – bowl-shaped depression located at the summit of the volcano that serves as the opening of the volcano to the Earth’s surface
- Secondary cone – smaller parasitic volcanoes that feed on the same magma chamber as the main volcano through secondary vents; usually emit volcanic gas called fumaroles
- Pyroclastic materials – any volcanic material that is extruded by a volcano, such as bombs, blocks, ashes, and others
Types of Volcano
a. Shield Volcanoes
Shield volcanoes are large dome-shaped volcanoes with broad gentle slopes and large craters. The largest volcano on Earth, Mauna Loa in Hawaii, is a shield volcano.
These volcanoes get their broad form due to the accumulation of layers of runny, fast-moving basaltic lava flows.
Shield volcanic eruptions are typically gentle and non-explosive, consisting of lava fountains, lava flows, and rarely any pyroclastic materials.
b. Cinder Cones

Cinder cones (scoria cones or ash-cinder cones) are steeper and have smaller craters than shield volcanoes. They are usually made up of loose pyroclastic material called scoria, a dark-colored igneous rock that is highly vesicular (has lots of vesicles or cavities) and made from extruded basaltic magma.
Cinder cone eruptions are moderately explosive, with lava coming from inside the vent or at the volcano’s base. Cinder cones usually have a short lifespan and are the most common type of volcanoes.
c. Composite Volcanoes or Stratovolcanoes

A composite volcano is probably the most recognizable form of the volcano, with its symmetrical steep-sided cone-shaped morphology. Alternating layers of viscous andesitic lava flows, volcanic ash, and cinders are responsible for their shape.
Eruptions tend to be violently explosive and can cause lava flows, pyroclastic flows, large ash clouds, and even lahar. Famous examples of stratovolcanoes are Mt. Fuji in Japan and Mt. Mayon in the Philippines.
When a particularly explosive eruption occurs, the stratovolcano could collapse, forming a large depression called a caldera. Our very own Taal Volcano is an excellent example of a caldera filled in by water, creating the Taal Lake.
Volcano-related Hazards
Volcanoes can be deadly forces of nature, and the impacts of volcanic hazards have been well-documented throughout the years. Here are some of the common volcanic hazards:
a. Pyroclastic Flow
A pyroclastic flow is a rapidly-moving current of hot gases and tephra (volcanic material) driven by gravity. They are also known as nuée ardentes (a French term meaning “glowing cloud”). Pyroclastic flows usually accompany explosive eruptions.
b. Lahars
Lahar flows occur when volcanic material becomes saturated with water, possibly from rainfall or melted ice, and rapidly descends steep volcano slopes.
This type of volcanic hazard is hazardous because it can happen even when a volcano is not erupting. The lahar flows during and after the 1991 Mt. Pinatubo Eruption is an excellent example of the destructive power that lahar flows can bring.
c. Lava flows
Depending on the viscosity of the lava, lava flows can spread out over large distances. Runny lava flows spread out more quickly before they solidify, compared to viscous lava.
Due to extreme temperatures (from 600°C to 1000°C), lava flows cannot be easily diverted or stopped. Fortunately, most lava flows can be outrun by a person on foot.
There are three main types of lava flows. The first one is called aa flows (pronounced as “ah-ah”) and is characterized by spiky and rough surfaces. The second one is called pahoehoe flows (pronounced as “pa-hoy-hoy”) and is described as having a “ropey” appearance with smooth surfaces. The last occurs when lava is extruded along the oceanic ridge, producing smooth rounded shapes called pillow lavas.
Volcanoes in the Philippines are classified as active (erupted within the last 600 years), potentially active, and inactive. As of 2020, there are 24 active volcanoes out of 407 volcanoes in the Philippines.
Module 10 Practice Test
11. Climate, Weather, and the Atmosphere
As previously discussed, the atmosphere is a collective layer of gas. The air that fills our atmosphere is composed of many different gases.
Components of the Atmosphere
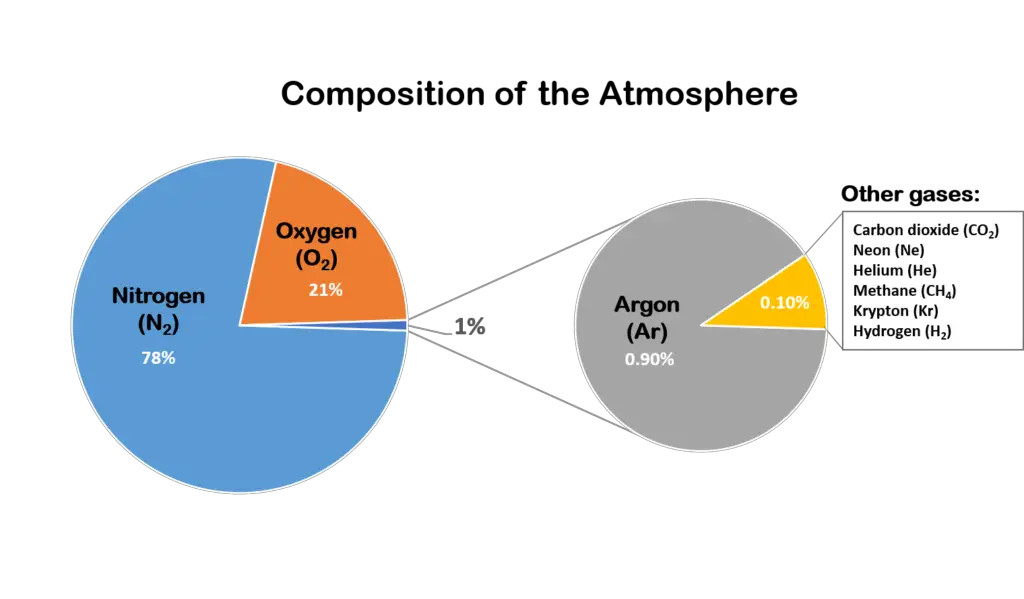
Besides these gases, the atmosphere is composed of minor and variable components such as water vapor, aerosols, and ozone that vary in abundance depending on time, location, and other factors.
Despite occurring in relatively small amounts, these components are still critical and can significantly affect the atmosphere.
a. Water Vapor
Water vapor is the primary source of precipitation and cloud formation in the atmosphere and, thus, a significant factor when predicting the weather.
Water vapor is also one of the most critical greenhouse gases because it helps absorb heat that radiates from the Earth, heating the atmosphere.
Greenhouse gases (GHG) trap heat in the Earth’s atmosphere. They include other gases such as carbon dioxide, methane, nitrous oxide, and ozone. Humidity refers to the amount of water vapor or moisture in the atmosphere.
b. Aerosols
These are minuscule solid and liquid particles that are suspended in the air. Aerosols include smoke, pollen, sea salt, dust, airborne microorganisms, and other natural or man-made sources.
Because of their size and weight, aerosols can remain suspended in the air for long periods of time (even years!). Aerosols have two crucial functions in the atmosphere:
- They can be “cloud seeds” or cloud condensation nuclei upon which clouds form, and
- They can also absorb, reflect, and scatter incoming solar radiation from the Sun, preventing harsh amounts of UV rays that can damage Earth’s inhabitants.
c. Ozone
As previously discussed, ozone is one of the atmosphere’s primary GHG (greenhouse gases). It is a form of oxygen with three oxygen atoms in each molecule (O3).
Like aerosols, ozone is important in absorbing potentially harmful UV radiation from the sun. Ozone depletion became a global issue in the 20th century primarily due to the overuse of chlorofluorocarbons (CFCs) that entered the atmosphere and broke down the ozone. This problem was addressed with the implementation of the Montreal Protocol by the United Nations to ban the production and use of CFCs starting in 1987.
Parts of the Atmosphere
The atmosphere is not uniform throughout. As you go from the bottom (Earth’s surface) to the top (toward space), you will observe changes in temperature and pressure.
As you go up the atmosphere, the pressure decreases due to fewer air molecules “pressing down” on you. Most air molecules are heavy and concentrated near the Earth’s surface.
On the other hand, temperature changes differently as you go from one atmospheric layer to another. Let’s take a look at the different layers of the atmosphere.
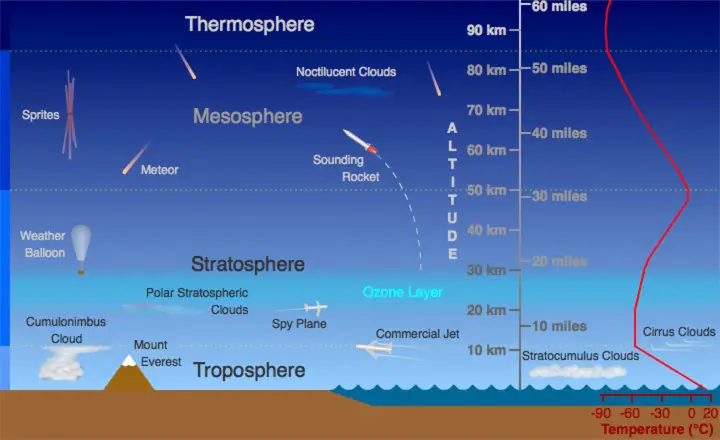
a. Troposphere
This is the lowest layer of the atmosphere. In this layer, temperature decreases with increasing altitude. The troposphere is the most crucial layer for meteorologists because all weather phenomena occur here.
The outermost boundary of the troposphere is called the tropopause.
b. Stratosphere
The temperature in this region increases with altitude because the ozone layer is located here.
As discussed earlier, the ozone layer becomes hot due to the absorption and trapping of UV rays from the Sun. Commercial airplanes fly in the lower portions of the stratosphere because of the less frequent turbulence experienced, unlike in the troposphere.
The end of the stratosphere is marked by stratopause.
c. Mesosphere
The coldest temperatures in the atmosphere (around -90°C) can be found at the end of this layer at the mesopause. The mesosphere protects us from meteors by burning up most meteors and asteroids before they reach the Earth’s surface.
d. Thermosphere
Temperatures start to rise again in this layer due to oxygen and nitrogen atoms’ constant absorption of high-energy radiation from the Sun. It is in this layer where satellites orbit around the Earth.
At the end of the thermosphere is a very thin layer of air called the exosphere which is considered the “final frontier” of the atmosphere.
Weather versus Climate
As mentioned above, the troposphere is where all weather phenomena occur. The study of weather phenomena is called meteorology.
Weather refers to the conditions of the atmosphere in a region over a short period of time. On the other hand, climate is the long-term behavior of the atmosphere over a region.
Meteorologists use humidity, air pressure, temperature, wind, and other factors to understand a region’s weather and climate better.
The Hydrological Cycle
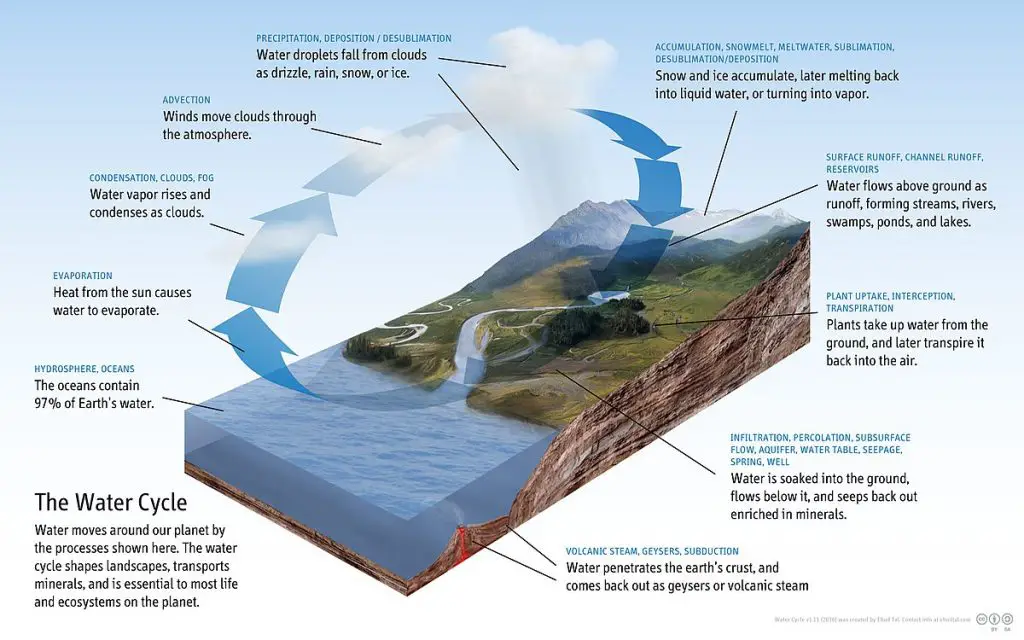
Water is inexplicably tied to many Earth processes, including processes in the atmosphere.
Water goes through a constant journey of evaporation and condensation called the hydrological cycle or water cycle, primarily driven by the radiation from the Sun. Regardless of what we can observe on the Earth’s surface, the hydrological cycle occurs continuously for millions of years above, on, and below the ground.
Although the water cycle does not have a starting point, we can start in the ocean, where the heat from the Sun evaporates water into vapor. Due to rising air currents, these water vapors are transported into the atmosphere, forming clouds.
Colder temperatures in the atmosphere encourage clouds to condense and precipitate. Precipitation reaches the surface of the Earth and flows down slopes as runoff. Some of the water seeps into the ground and replenishes the groundwater in aquifers (underground freshwater reservoirs).
Eventually, all rivers and streams arrive at their ultimate destination, the ocean, and the cycle repeats.
Indeed, the hydrological cycle is an essential system on which all Earth’s residents depend.
Cloud Formation
Clouds are one of the easily observable indicators of weather conditions.
Clouds start when water vapor in the air changes to liquid in condensation and forms around a “cloud seed” or condensation nuclei (aerosols). Soon, a cloud is formed from millions of tiny cloud droplets.
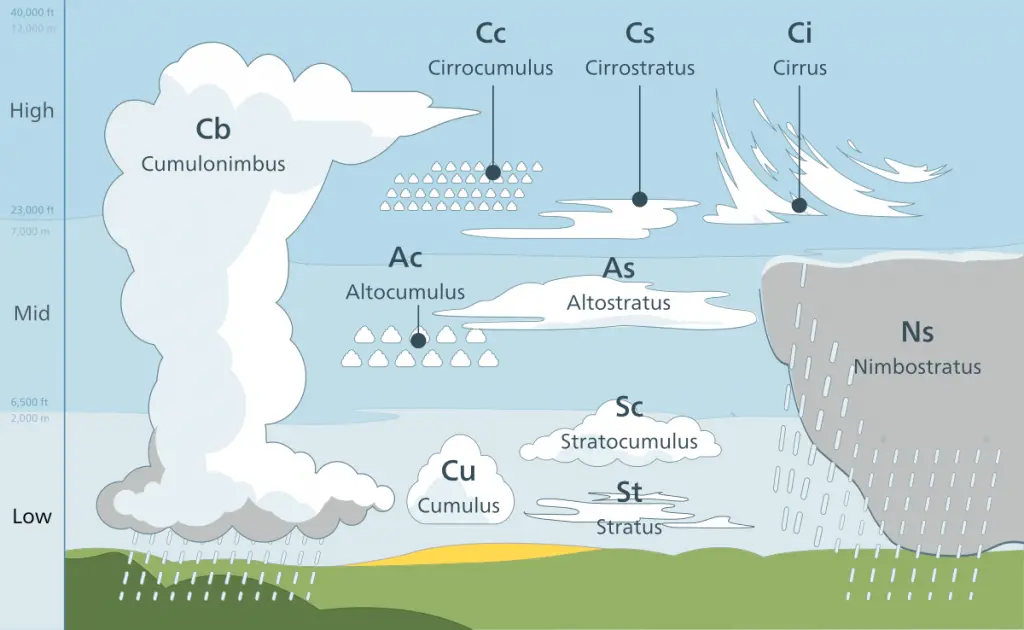
Different types of clouds are classified according to form and height. Based on height, there are low clouds (0-2000 m), middle clouds (2000-6000 m), and high clouds (over 6000 m).
There are three main types of clouds based on the form:
- Cirrus clouds (From the Latin word cirrus meaning “lock of hair”). These are thin, wispy, and white clouds that resemble hair.
- Stratus clouds (From the Latin word stratum meaning “layer”). These are thin layers of clouds that cover extensive portions of the sky.
- Cumulus clouds (From the Latin word cumulo meaning “a heap”). These are big, cotton candy-looking clouds that can stack vertically in a tower-like manner.
Combinations of these primary forms result in the formation of different cloud types.
Wind Formation
The wind is generated when air flows from regions of high pressure to regions of low pressure caused by the unequal heating of the Earth’s surface. The following factors control it:
a. Pressure Gradient Force
Physics tells us that when an object encounters an unbalanced force in one direction, it will accelerate in the same direction. This is what happens when there are horizontal pressure differences in the air. This variation in air pressure is the driving force of the wind.
b. Coriolis Effect
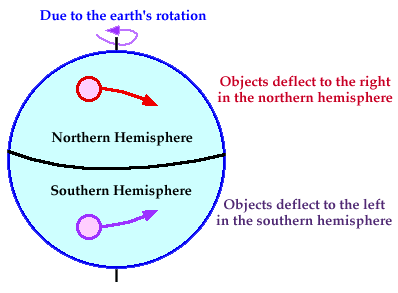
When the wind moves, it does not go in a straight line. It is deflected from its original path due to the Earth’s rotation in a phenomenon called the Coriolis Effect.
The Earth spins counterclockwise, so all free-moving objects (including the wind) are deflected to the right in the Northern Hemisphere and the left in the Southern Hemisphere.
c. Friction
Friction with the Earth’s surface is caused by the terrain the winds encounter. This could include mountains, hills, forests, and even man-made structures that hinder wind flow.
Other Types of Weather Phenomena
a. Typhoons, hurricanes, and cyclones
These refer to the same thing: areas of low pressure that form over oceans characterized by a spiral movement of viral winds. The only difference between them is where they formed.
Typhoons are storms that form in the Western Pacific. Hurricanes are storms in the Atlantic and Eastern Pacific, while cyclones form over the South Pacific and the Indian Ocean.
b. Thunderstorms
These are associated with cumulonimbus clouds, heavy rainfall, thunder, lightning, and sometimes tornadoes. The upward movement of moist and warm air causes them.
Lightning is caused by the electric charge that results from the collision of ice crystals (cloud droplets) in the air.
c. Tornadoes
These are columns of violently spinning air that extend downwards from cumulonimbus clouds. Most tornadoes are short-lived but can still cause extensive damage to property, nature, and life along their path.
d. Precipitation
Precipitation occurs when any form of water particle descends from the atmosphere toward the Earth’s surface. The most common form of precipitation is rain (water droplets). Other types of precipitation can include sleet (pellets of ice), hail (lumps of ice), snow (ice crystals), and drizzle (very fine water droplets).
e. El Niño
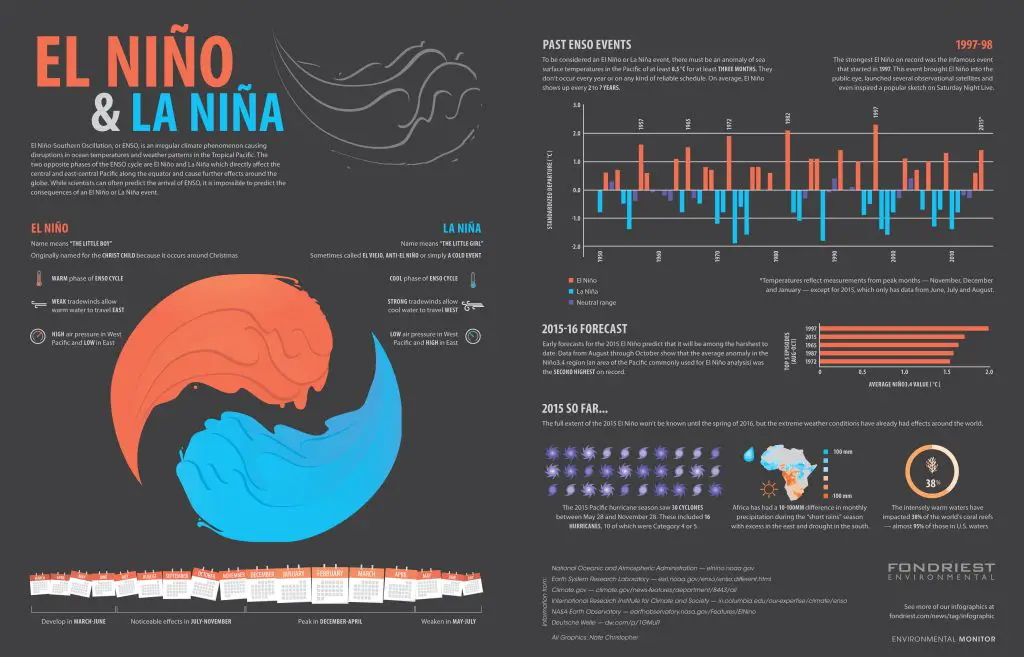
This is a weather pattern that affects countries near the Southern Pacific Ocean.
During normal conditions, wind along the equator pushes warm surface water near South America towards the Western Pacific countries (such as Indonesia, Philippines, etc.). The warm surface water is soon replaced by cooler water from underneath.
However, during El Niño, warm water from the Western Pacific flows instead towards South America and up north towards the western portion of North America. This phenomenon induces changes in weather patterns, marine fisheries, and ocean conditions.
El Niño is part of a weather cycle called El Niño-Southern Oscillation (ENSO) and is known as the warm phase of ENSO. La Niña, the cold phase, can be considered the opposite of El Niño.
The Philippine Atmospheric, Geophysical, and Astronomical Services Administration (PAGASA) is the authority on all meteorological, climatological, and astronomical phenomena related to the Philippines. PAGASA monitors tropical cyclone activity within the Philippine Area of Responsibility (PAR).
Module 11 Practice Test
12. A Brief History of Astronomy
Everything and anything on Earth is part of the Universe, a vast expanse filled with planets, stars, moons, comets, galaxies, black holes, and other celestial bodies we have yet to discover.
Our Earth is just a speck of dust in one galaxy out of billions of galaxies. Nevertheless, human curiosity knows no bounds. So to better understand our place in this Universe, we turn to astronomy, the study of celestial objects and phenomena in space.
Early Astronomy
The roots of modern astronomy or early astronomy can be traced back to Ancient Greece during the “Golden Age” of astronomy (600 B.C. – 150 A.D.). During this time, the early Greeks developed geometry and trigonometry and utilized these to discern and describe observable celestial bodies.
The early Greeks believed that the Earth was the center of the universe and that all the other heavenly bodies orbited around it. This was known as the geocentric model (from the Greek word geo, meaning “Earth”) of the universe.
And although great minds like Aristarchus (312-230 BC) also offered the possibility of a heliocentric model (from the Greek word helios meaning “Sun”) of the universe, which suggested that the Earth and other planets revolved around the Sun, the idea would not gain traction until the 15th century.
Despite this incorrect belief, early Greeks still contributed many essential discoveries in the name of astronomy.
Here are some of the most notable contributors to early astronomy:
As early as the 5th century BC, Greek philosophers such as Parmenides believed in a spherical Earth, but this belief was purely based on philosophical assumptions.
However, Aristotle (384-322 BC) would give scientific credibility to this idea by observing the shape of the Earth’s shadow that is cast upon the moon during eclipses. Since the shadow cast on the moon is round, the Earth would have to be spherical, not flat or disk-like, to produce such a shape.
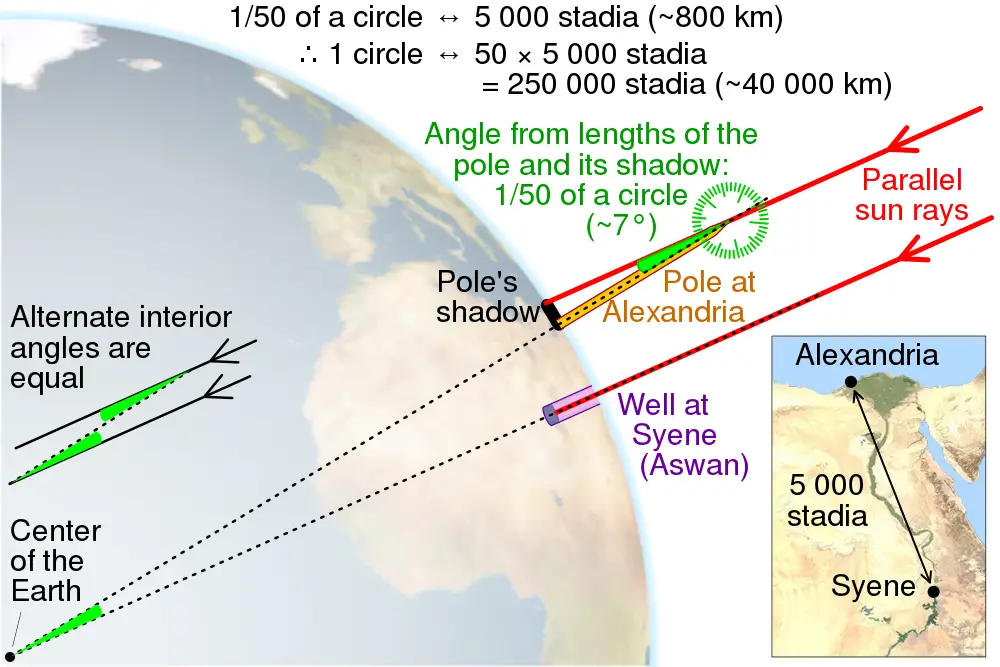
Eratosthenes (276 – 194 BC) is credited for successfully establishing the circumference of the Earth by observing the angles of the Sun’s rays at noon in two Egyptian cities. By discovering that the angles of the rays differed by several degrees, he calculated that the circumference of the Earth is around 39,400 km, close to the actual modern-day value of 40,075 km.
Perhaps one of the most significant early Greek astronomers is Hipparchus (190 – 120 BC), known for his significant contributions to the field of astronomy: the development of trigonometry, accurately estimating the distance between the Moon and Earth, near accurate estimation of the length of a year, and a star catalog of nearly 850 stars classified according to their brightness (adapted into the Hertzsprung-Russell diagram).
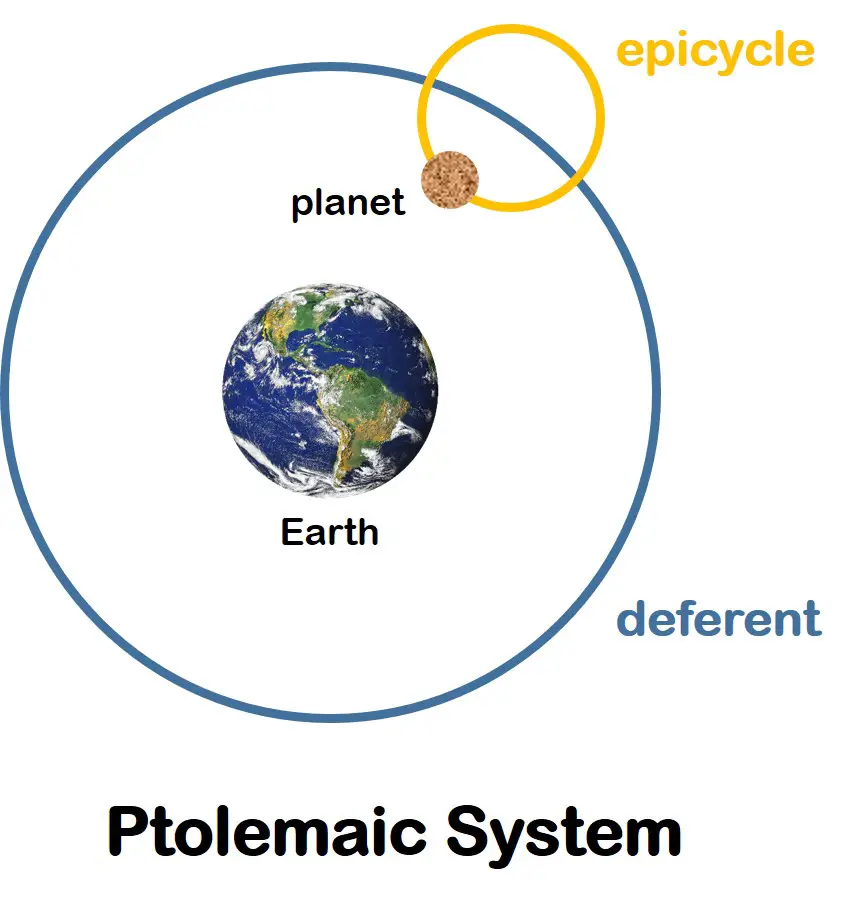
Claudius Ptolemy (100 – 170 AD) developed the Ptolemaic system, a geocentric model of the universe accounting for the apparent motion of the planets as it revolves around a stationary Earth. Despite using an incorrect model, he could still predict the planets’ positions using a combination of large circles (deferents) and small circles (epicycles) to represent the planets’ orbits.
Even with the decline of the Roman Empire, the Ptolemaic system became the prevalent model of the universe for several years.
The Emergence of Modern Astronomy
The center of astronomical study would shift to Baghdad, Iraq, after the fall of the Roman Empire during the 4th century.
Arabic astronomers translated the works of Greek astronomers and expanded them through their own works. Centuries would pass before astronomy was reintroduced to Europe through interaction and trade with the Arabic community. Scientific thought started to break away from religion and philosophy during this period.
Here are some of the most notable astronomers and scientists that contributed to the development of modern astronomy:
Nicolaus Copernicus (1473 – 1543) was a Polish astronomer who advocated for a heliocentric universe model, later called the Copernican system, after discovering Aristarchus’ works. Even though his model was more correct than Ptolemy’s, Copernicus could not account for planetary motion. His idea that the Earth was not the center of the universe was considered heretical then and, thus, met with much criticism.
Johannes Kepler (1571 – 1630) was a German astronomer who served as an assistant to Tycho Brahe (1546 – 1601), a Danish astronomer. With an observatory at their disposal, Brahe and Kepler observed and measured the locations of different celestial bodies. Tycho did this to refute the Copernican system. Despite serving under Brahe, Kepler remained steadfast in his belief in the Copernican model. After Tycho’s death, Kepler used the data they gathered to formulate the fundamental laws of planetary motion:
The Three Basic Laws of Planetary Motion
1. All the planets move around the Sun in an elliptical orbit, not circular as previously believed. This is also known as the Law of Ellipses.

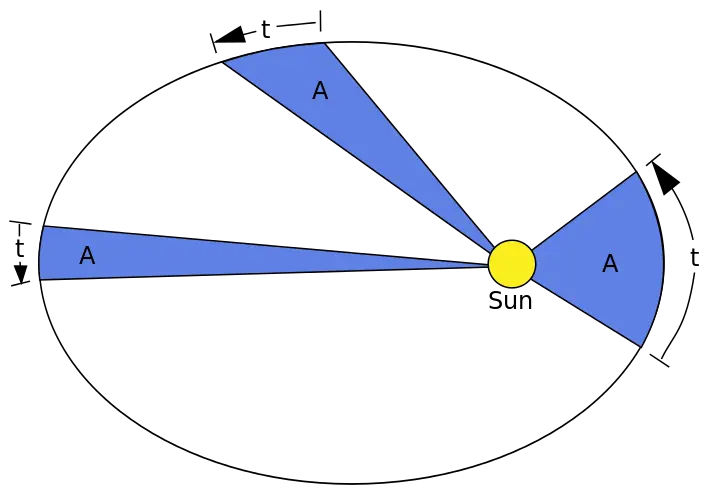
2. If you trace an imaginary line from a point in the orbit to the Sun as a planet revolves, the line sweeps over equal areas in equal time intervals. This is also known as the Law of Equal Areas and explains the variation in the speed at which planets orbit around the Sun. The perihelion refers to a point where the planet’s orbit is closest to the Sun, while the aphelion refers to when it is farthest.
3. The square of a planet’s orbital period is proportional to the cube of a planet’s mean distance to the sun. This is also known as the Law of Harmonies.
Despite understanding how planets move, Kepler could not establish why they moved that way and what keeps planets in their orbits instead of floating away into space.
Galileo Galilei (1564 – 1642) was an Italian astronomer, a contemporary of Kepler, and a supporter of the Copernican model. He built several telescopes, which aided him in making more detailed observations of heavenly bodies. He observed the surface of the Moon, which was previously believed to have been smooth as glass but has now proven to contain craters, mountains, and plains like the Earth. He discovered that Jupiter had four moons that revolved around it, further dispelling the notion that the Earth was the center of motion in the universe. He also discovered the existence of sunspots, relatively darker and cooler areas on the surface of the Sun. Unfortunately, prolonged observation of the Sun damaged his eyesight and eventually completely blinded him.
Sir Isaac Newton (1642 – 1727) was a prominent English scientist who significantly contributed to math, physics, and astronomy. One of his most notable contributions was the answer to the question of what kept planets in orbit: the Law of Universal Gravitation. According to the law, everybody in the universe attracts every other body with a force proportional to the mass of the bodies and inversely proportional to the square of the distance between the bodies.
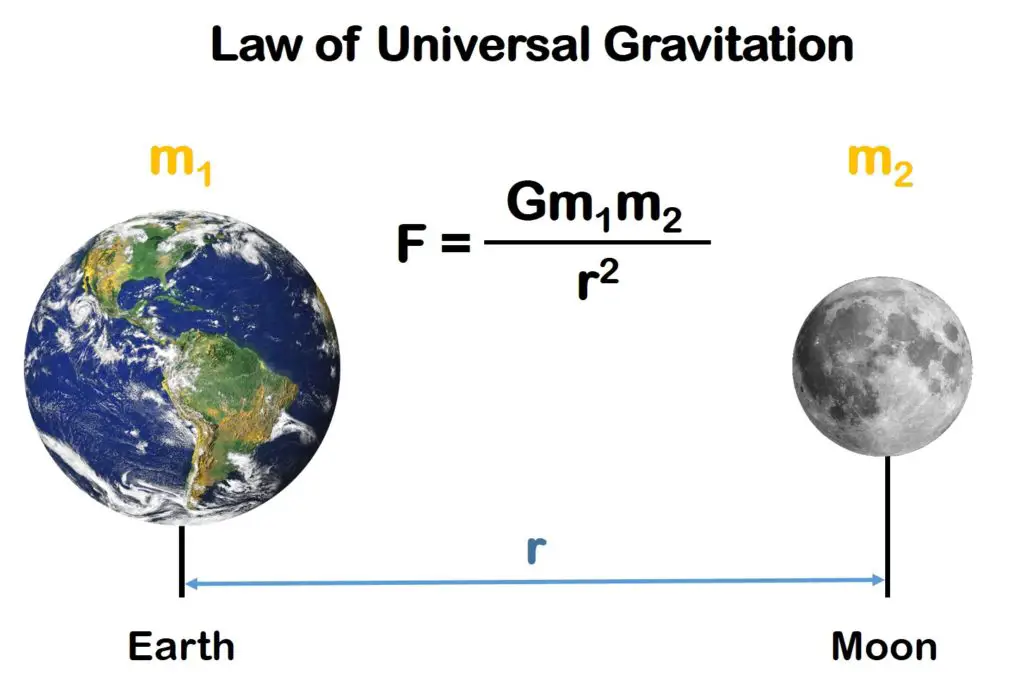
According to the equation, the larger the mass of an object, the bigger the gravitational force it exerts. This would explain how the Moon affects the tides on Earth and supports the idea that the massive Sun is the center of our system.
The force of gravity and the tendency of a planet to move in a straight line contribute to the orbit of a planet around the Sun. Without the pull of gravity, the planets would move forward in a straight line out into space. Without the tendency of the planet to move in a straight line, the planets would fall into the Sun because of its immense gravitational pull.
Module 12 Practice Test
13. The Origin of the Universe and the Solar System
Cosmology is the study of the origin and evolution of the universe. In this module, we’ll focus on the basics of cosmology to explore the theories explaining how our universe and the solar system formed billions of years ago.
The Big Bang Theory: Origin of the Universe
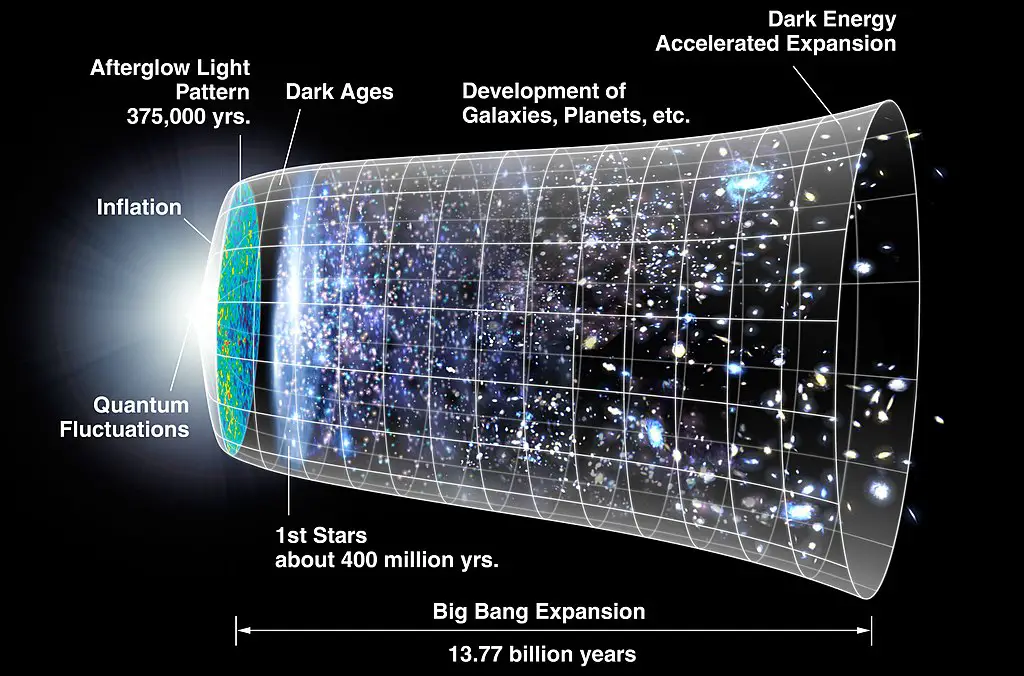
The Big Bang Theory (BBT) is currently the most accepted theory explaining the universe’s origin.
According to the BBT, before the universe (or anything and everything) existed, all matter and energy were condensed into high-temperature and high-density states. Suddenly, rapid expansion occurred, resulting in an explosion (hence the name, Big Bang), which generated all matter and energy, including space and time.
After the explosion, the Universe was in a hot and dense state. As expansion continued, the temperatures cooled down and allowed for the formation of light elements such as hydrogen and helium. The energy was readily available and encouraged nuclear fusion, generating other slightly heavier elements.
Due to the force of gravity, clumps of matter began to form and grow through accretion. Accretion would eventually produce large masses of matter, forming into the first stars 400 million years after the Big Bang.
A supernova occurs at the end of a star’s life cycle.
Supernovas occur when a star’s core collapses and generates spectacularly powerful explosions. They are the primary source of heavier elements such as nickel and iron. Large masses eventually form into planets, asteroids, and other space bodies. Gravity would also affect these space bodies and come together to form galaxies.
Based on the joint effort of NASA and ESA, the most accepted age of the Universe is 13.8 billion years old. They determined this by measuring the leftover radiation from the Big Bang called cosmic microwave background (CMB).
Even though the universe is billions of years old already, expansion has not stopped and persists. This is what Edwin Hubble, an American astronomer, observed. He noted that galaxies seemed to be moving away from each other and did so with incredible velocity.
He also noted that galaxies farther away from the Earth move away at a more incredible velocity. By observing the visible light spectra, he noted that galaxies moving away from us produce a redshift, meaning the wavelength becomes longer. This phenomenon differs from when something moves towards us, producing a blueshift or shorter wavelength.
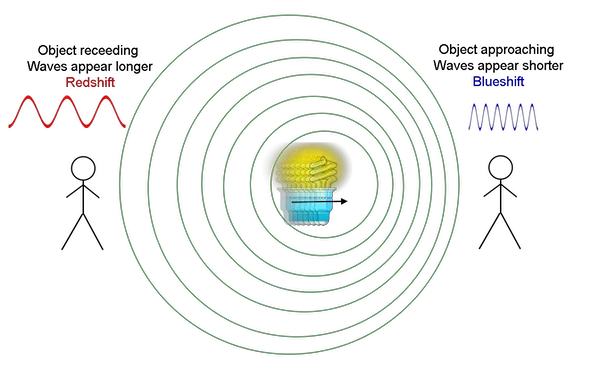
A famous thought experiment used to understand this is the Doppler Effect: Imagine an ambulance passing by. The siren’s pitch lowers as the ambulance approaches you, meaning the wavelength is getting longer. As it passes you, the siren’s pitch becomes higher, signaling that the wavelength is becoming shorter.
The Nebular Theory: Origin of the Solar System
The Nebular Theory was proposed and developed by several proponents (namely Emanual Swedenborg, Immanuel Kant, Pierre-Simon Laplace, and Victor Safronov) throughout the years until it became the accepted model of the origin of the solar system in the 20th century.
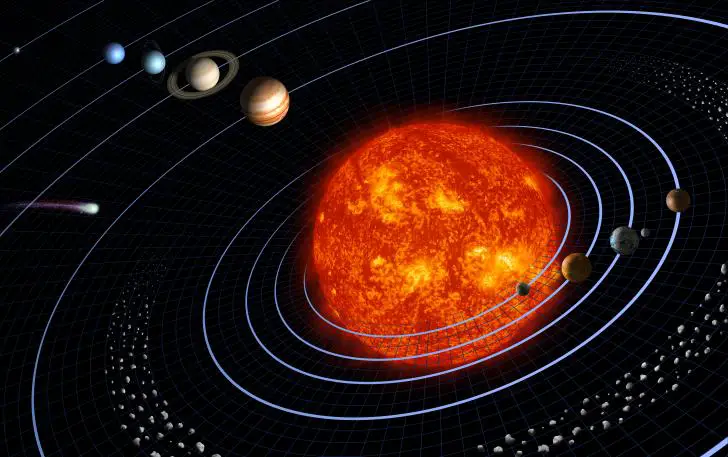
According to the theory, our solar system formed from an enormous rotating cloud of dust and gas called the solar nebula nearly 4.6 billion years ago.
Over time, the solar nebula began to contract and flatten due to gravity. As contraction continues, gravitational energy is converted into thermal energy, generating high temperatures. High temperatures and the inward pull of gravity in the center of the cloud led to the formation of a proto-Sun.
As contraction declined, the temperatures began to cool, condensing rock-forming elements such as Fe, Ni, Si, Ca, Na, and others. Particles began to collide and accrete into masses called planetesimals (proto-planets) which eventually formed planets.
Due to higher temperatures within the center of the solar system, heavier rock-forming elements condensed to form the inner planets (terrestrial planets). Planets within the outer ring of the solar system are called inner planets (jovian or gaseous planets) and are formed with small, rocky cores but are primarily composed of ice and gas such as CO2, H2O, NH3, and CH4.
Module 13 Practice Test
14. The Solar System and Its Planets
Our Solar System is part of the Milky Way Galaxy. According to NASA, it currently consists of the Sun, eight planets, five dwarf planets, and more than 200 moons.
The first four planets from the Sun are called the inner planets. They are separated from the outer planets by the asteroid belt, a region between Mars and Jupiter primarily composed of irregularly shaped and sized asteroids.
Let’s discuss all these celestial bodies one by one.
The Sun

The Sun is a yellow dwarf star and the center of our solar system. It is so massive that it constitutes nearly 99.8% of all the mass in the solar system, while 0.2% is only represented by the planets.
It is 4.6 billion years old, as old as our solar system. It is a big ball of gas composed of 92.1% hydrogen and 7.8% helium. The surface of the Sun is the coolest portion with temperatures of 5,600℃. The hottest temperatures can be observed in the core, where temperatures can go as high as 15,000,000℃.
Distances in the solar system are based on the distance between the Sun and the Earth, called 1 astronomical unit (AU), which is equivalent to 150 million km.
The Terrestrial Planets
a. Mercury

Mercury is the innermost planet closest to the Sun and the smallest of all planets, slightly larger than the moon. It is the second densest planet after the Earth.
Despite being closest to the Sun, it is not the hottest planet in the solar system (that distinction belongs to Venus). Temperature changes during daytime and nighttime are extreme, with temperatures reaching 430℃ during the day and -180℃ at night. It has no known moons.
b. Venus

Venus is the second planet from the Sun and the hottest planet in the solar system. Out of all the atmosphere-bearing planets, it has the densest atmosphere composed primarily of CO2.
Venus spins in the opposite direction (retrograde motion) compared to other planets. The longest-known lava channel in the solar system, Baltis Vallis, can be found here. It has no known moons.
c. Earth

Earth is the third planet from the Sun and our home planet. As far as we know, it is the only planet in the solar system suitable for life due to the presence of liquid water on the surface. It has one moon, aptly named Moon. We will discuss the formation of the Earth and how it moves in more detail in the next module.
d. Mars

Mars is the fourth planet from the Sun and our closest neighbor. It is said that out of all the planets, Mars is the closest to being Earth-like.
After the Earth, it is the second most explored planet in the solar system. It is also known as the “red planet” because of the iron oxide in its rocks, giving it a reddish hue.
The largest volcano in the solar system, Olympus Mons, can be found here. Expeditions on the surface of Mars showed evidence of the presence of water in the planet’s early years. It has two known moons, Phobos and Deimos.
The Jovian Planets
e. Jupiter

Jupiter is the fifth and largest planet in the solar system and the largest of the four gas giants. It is mostly composed of gas and has a small solid inner core the size of Earth.
The colorful bands seen on Jupiter’s surface result from the convective gas flow. The light-colored areas represent warm, ascending material, while the dark-colored areas represent the cold, sinking material.
The Great Red Spot is a noticeable area on Jupiter’s surface, representing the largest storm on the planet and the solar system. Jupiter has more than 75 known moons and a ring system.
f. Saturn

Saturn is the sixth and second-largest planet from the Sun. It also exhibits the most spectacular ring system of all the gas giants.
Its seven rings and thousands of ringlets comprise rocks, ice, and other space debris. Like Jupiter, it is mainly composed of gas and has a small solid rocky core.
Aside from its ring system, it has 62 known moons and possibly more.
g. Uranus

Uranus is the seventh planet from the Sun and is uniquely known as the “sideways planet” because of the sideways orientation of its axis, as opposed to the rest of the planets.
As a gas giant, it has a small rocky core mostly made of gas and ice. It was the first planet to be discovered using a telescope in the 18th century. Although not as spectacular as Saturn’s, it has a ring system and over 27 known moons.
h. Neptune

Neptune is the farthest planet from the Sun. As we all know by now, it is a gas giant and, therefore, also has a rocky core and primarily gaseous composition.
It is known as the “windiest planet” in the solar system, with wind speeds of up to 2,000 km/h. Like Jupiter, it has dark spots named Great Dark Spots where raging rotating storms occur. However, they are more short-lived than Jupiter’s Great Red Spot.
It also has a ring system and around 14 known moons. Beyond Neptune is a vast donut-shaped region containing icy bodies called the Kuiper Belt. The icy bodies are Kuiper Belt Objects (KBOs) or trans-Neptunian objects (TNOs). Some of these icy bodies are considered dwarf planets, which we will discuss in more detail below.
The Dwarf Planets

According to the International Astronomical Union (IAU), to be classified as a planet, a celestial body must:
- Be in orbit around the Sun
- Have sufficient mass to assume hydrostatic equilibrium (Its gravitational pull must be strong enough to assume a rounded shape)
- Have cleared the neighborhood around its orbit (It has to be the dominant gravitational body in its orbit and either attract or push away smaller bodies intersecting its orbit)
Celestial bodies that pass one or two conditions but fail the rest are called dwarf planets. As of 2020, there are five known dwarf planets.
- Pluto – Pluto was considered the 9th planet until it was stripped of its planet status in 2006 because it could not satisfy the third requirement–-it did not “clear the neighborhood” as it revolved around the Sun. Its size is large enough for its gravitational force to maintain five known moons. It is located in the Kuiper Belt.
- Eris – Eris’s diameter is nearly the same as Pluto’s, and they are considered the two largest dwarf planets. It is located nearly three times farther than Pluto, extending beyond the Kuiper Belt. Before being named Eris, it was named 2003 UB313.
- Ceres – is the largest object in the asteroid belt that separates the inner and outer planets. It is also the smallest and the earliest known dwarf planet, being discovered in 1801.
- Haumea – is a football-shaped dwarf planet in the Kuiper Belt beyond Neptune. Its distorted shape is attributed to its fast spin; it is considered one of the fastest-rotating objects in our solar system.
- Makemake – Like Haumea, Makemake can be found along the Kuiper Belt. Before being designated as a dwarf planet, it was named 2005 FY9.
Module 14 Practice Test
15. How the Earth Was Formed
Earth started 4.6 billion years ago from planetesimals that formed through accretion and gradually formed into planets. This is known as the core accretion model, where the Earth started.

The early Earth was dominantly molten because of the constant bombardment of space bodies and the decay of radioactive elements. As time passed, bombardment declined, and the temperatures began to cool.
How Earth Formed Its Layers
Heavier elements like iron and nickel sank to the center of the Earth, forming the core, while lighter elements migrated towards the surface. This allowed the Earth to produce layers in a process called chemical differentiation. During this cooling period, the Earth’s primitive crust started to form, and the magnetic field was produced.
How the Moon Formed and What Caused the Tilt in the Earth’s Axis
The Giant Impact Hypothesis states that around 4.5 billion years ago, a large Mars-sized celestial body called Theia impacted the Earth, resulting in the ejection of undifferentiated Earth materials, forming our Moon.
The impact is also said to have been the cause of the tilt in the Earth’s axis.
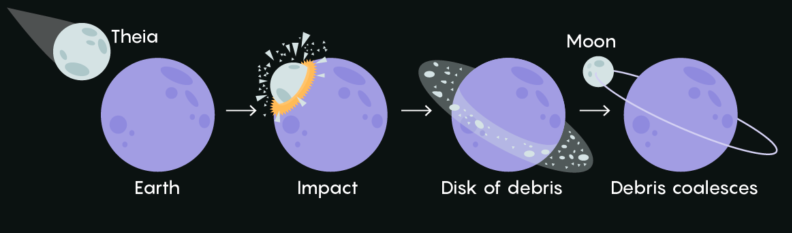
How Life on Earth Started
Around 4.37 – 4.20 billion years ago, Earth was bombarded by asteroids during the Late Heavy Bombardment stage. It is theorized that these asteroids carried atmospheric, oceanic, and biological components such as water and bio-elements, which set the stage for life on Earth.
As the Earth cooled, the outgassing of gases (via intense volcanism) from the Earth’s interior formed Earth’s first atmosphere. Unlike now, the first atmosphere was very hot and mainly composed of gases such as NH3, CH4, CO2, and some H2O.
Around 4.0 billion years ago, atmospheric water vapor began to condense, producing torrential rains forming our vast oceans.
Cyanobacteria (aerobic bacteria) were essential in producing and increasing O2 in the Earth’s atmosphere. Because of this, aerobic bacteria began to thrive, while anaerobic bacteria declined in the Great Oxidation Event.
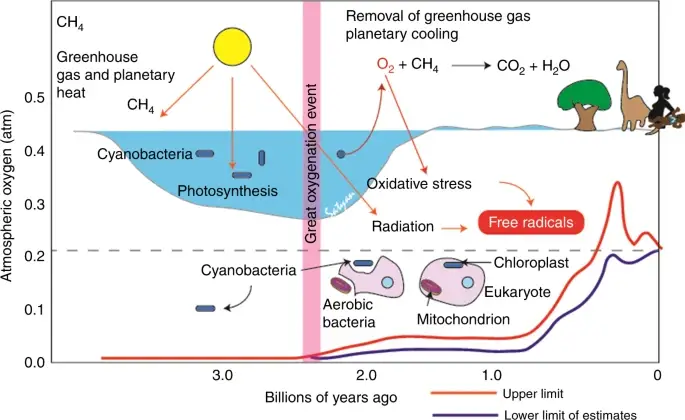
Accelerated weathering of the Earth’s surface introduced elements such as Na, Ca, K, and Si from the land to the oceans, increasing its salinity. Seawater and CaCO3 in the oceans “locked up” large amounts of CO2 in the atmosphere, cooling the Earth significantly.
Module 15 Practice Test
16. The Motions of the Earth
The Earth goes through three types of motions: rotation, revolution, and precession. Let’s review them one by one.
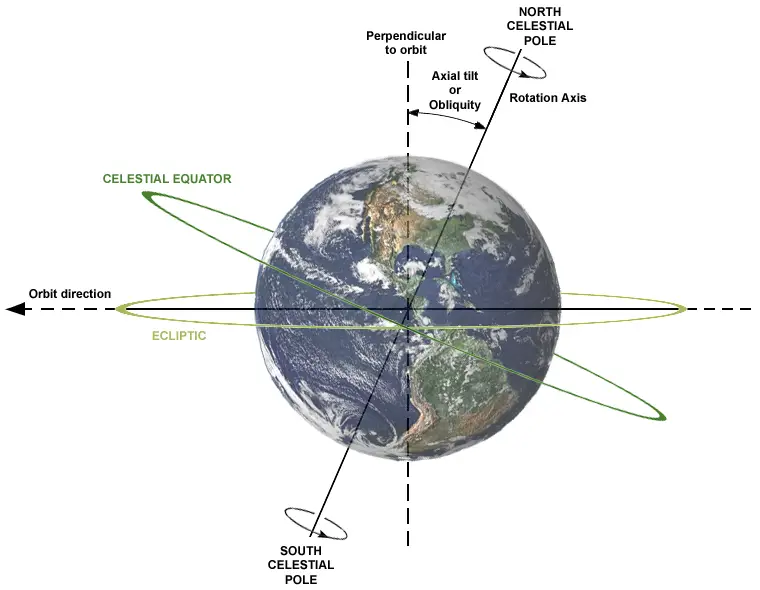
Earth Rotation
The rotation occurs when a body, such as the Earth, spins on its axis. The axis is an imaginary line that passes through the center of the Earth, going through the North Pole and exiting through the South Pole. It is what gives us night and day.
One complete Earth rotation equals 23 hours, 56 minutes, and 4 seconds. The Earth’s axis is not perpendicular to its orbital plane. Instead, it is tilted 23.5 degrees. However, the value changes as the axial tilt varies between 22.1 to 24.5 every 41,000 years (one Milankovitch Cycle). The change in the axial tilt is called obliquity.
Because of the Earth’s axial tilt, different regions experience varying intensities of seasons and different lengths of daytime. The winter solstice is the shortest day of the year, while the summer solstice is the longest. An equinox occurs when day and night are equal in length.
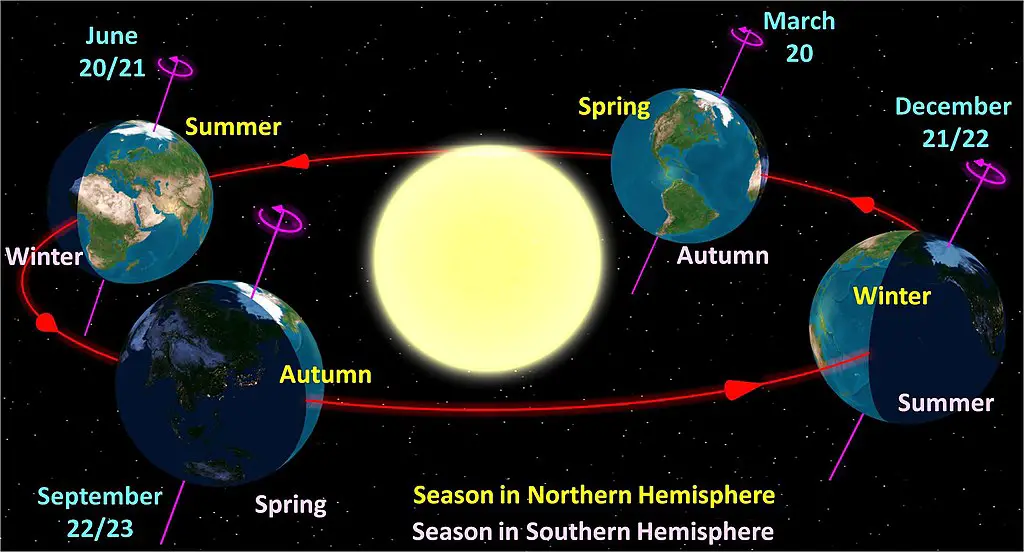
Earth Revolution
As the Earth rotates, it orbits around the Sun in a process called revolution. One complete Earth revolution requires 365 days, 6 hours, and 9 minutes at an average speed of 30 km/s.
When the Earth is at its aphelion, its position in its orbit is farthest from the Sun. Earth is closest to the Sun when it is at its perihelion.
The Earth experiences a third type of motion that is slower and less prominent– precession. Axial precession represents the “wobble” of the Earth as it rotates on its axis, much like a spinning top. Throughout 26,000 years, the direction in which the axis points changes until it completes 360 degrees.

Module 16 Practice Test
17. The Motions of the Moon
The Moon is the Earth’s only natural satellite. It completes one revolution around the Earth every sidereal month or 27.32 days. However, it takes 29.5 days or one synodic month for the Moon to cycle through its phases.
Phases of the Moon
Every month, we observe the Moon go through eight phases. The different phases result from the Moon’s motion and the amount of sunlight reflected off the Moon’s surface.
As we start from the new moon, more portions of the moon can be seen (waxing). When it reaches the full moon phase, portions of the moon start to decrease in visibility (waning) until it reaches the new moon phase again.

Eclipses
As the Earth revolves around the Sun and the moon revolves around the Earth, certain positions will allow for eclipses to happen.
A lunar eclipse occurs when the Earth comes between the Moon and the Sun, blocking any sunlight from reaching the Moon’s surface. This happens when the Moon falls within the inner shadow cast by the Earth called the umbra. The penumbra is the outer portion of the Earth’s shadow that only partially blocks the sun’s rays. On the other hand, a solar eclipse occurs when the Moon comes between the Sun and the Earth and casts a shadow on the Earth.
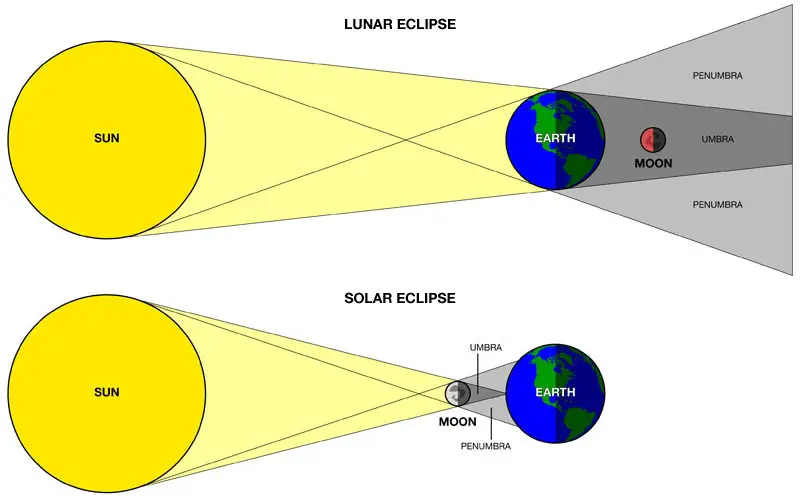
Eclipses are rare because the Moon’s orbit is tilted approximately 5℃. Eclipses can only occur when a full moon or a new moon occurs while the Moon’s orbit crosses the plane of the ecliptic. Because of this, the usual number of eclipses per year is four.
Module 17 Practice Test
18. Other Celestial Bodies in Space
Given the vastness of the universe, it is no surprise to find that we have other celestial neighbors besides other planets.
a. Stars
Stars are considered the building blocks of galaxies. Without stars, elements essential to the formation of life would not exist.
Much like the formation of the Sun, stars form from interstellar clouds of dust and gas that condense due to gravitational pull.
Nuclear fusion is the driving force behind the life of a star. At the very core of a star, hydrogen undergoes nuclear fusion to form helium, producing an outflow of energy that keeps a star from collapsing under its own weight.
A star nears the end of its life when it exhausts its fuel supply, leading to collapse and then a spectacular explosion in the form of a supernova. The materials produced by a supernova may be reused again in the formation of another star, repeating the cycle.
Stars are classified according to the relationship between their absolute luminosities or magnitudes and temperatures in a diagram called the Hertzsprung-Russell diagram (HR diagram). Depending on the star, it may be plotted along the middle diagonal line called the Main Sequence or fall above it (giants and supergiants) or below it (white dwarfs).
As mentioned, our Sun falls on the Main Sequence and is classified as a Yellow Dwarf.
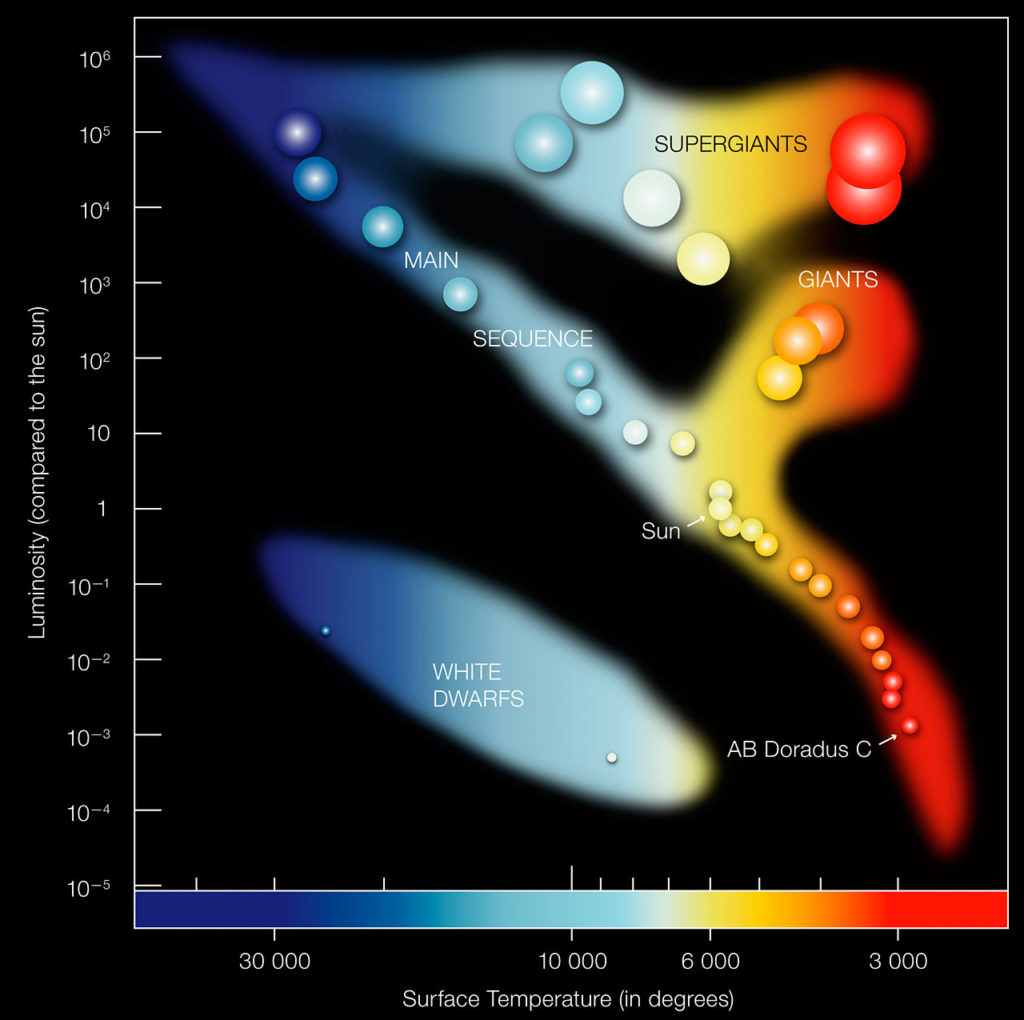
b. Comets

Comets are small space bodies of dust, ice, and frozen gases. When comets come close to the Sun, they heat up and release gas through outgassing, leaving long trails of gas and dust as it moves.
Comets have different orbital periods (the time it takes for a comet to complete one orbit around the Sun). Some famous comets are Halley’s Comet (76-year orbital period), Comet Hale-Bopp (2,533-year orbital period), and Comet Hyakutake (113,782-year orbital period).
c. Asteroids

These are small rocky bodies (planetesimals) that are leftovers from the formation of the Solar System. Most asteroids in the solar system can be found in the asteroid belt region. They are irregularly shaped and greatly vary in diameter.
When located in space, they are called asteroids (when more than one meter in diameter) or meteoroids (when less than a meter in diameter). When it enters the Earth’s atmosphere, it becomes a meteor. It is called a meteorite if it reaches the Earth’s surface.
d. Galaxies

Galaxies are large clusters of stars, solar systems, gas, and dust held together by gravity. Using the Hubble Space Telescope, astronomers estimate that there may be 100 billion galaxies and probably more.
Galaxies are classified based on morphology and divided into three major categories: elliptical, spiral, and irregular. As mentioned in previous chapters, we live in the Milky Way, a spiral-type galaxy. However, many galaxies also appear in unusual forms, usually combining the main morphologies.
Galaxies tend to be bundled together in groups, clusters, and superclusters. For example, the Milky Way galaxy is part of the Local Group galaxy group, which is part of the Virgo Supercluster and the even greater Laniakea Supercluster.
e. Black holes

A black hole refers to a region in space where the force of gravity is strong enough that even light cannot escape.
This is the result of matter being compressed beyond a specific density. Blackholes are sometimes the result of extreme compression that happens when a star dies. Many galaxies are believed to contain massive black holes at their centers.
The Milky Way’s massive black hole is called the Sagittarius A* (pronounced as “Sag A star”).
Module 18 Practice Test
Earth Science Study Tips for Students and UPCAT/College Entrance Test Takers
1. Repetition is the key
The number of terms to be memorized in Earth Science can be daunting, and there’s really no way around that. My personal suggestion? Repetition.
After studying this reviewer, try re-writing the definition of a term or the
steps in a process in your own words. This will not only test your memory
but also your understanding of the topic.
2. Reinforce learning through flashcards
In addition to the tip above, try making notes differently: write
them down on paper, make flashcards, type them in a document, etc., to reinforce and retain the information much more effectively.
3. Read up on current and past events related to Earth science
UPCAT and other entrance exams sometimes include questions about significant natural events (e.g., earthquakes, eruptions, typhoons, etc.) in the Philippines. This tests your knowledge and social awareness of current and past issues. Before the examination, try to read up on news articles about recent events and know important historical ones (hint: things like the Mt. Pinatubo Eruption or the most recent volcanic eruption/s
may pop up!)
4. Master the concepts without relying on rote memorization
When you need to memorize a diagram like the hydrological or rock cycles, redraw it repeatedly until you master it. Don’t forget to understand what happens behind each step.
When studying topics that require you to memorize chronological events (like the Big Bang Theory or the Nebula Theory), try teaching the topic to someone (a friend, a sibling, or even a stuffed toy) like you’re trying to tell a story. It’s very effective, and you could also teach someone something new!
For visual learners, here are some videos that will help you remember certain Earth science concepts and processes:
- Rock Cycle
- Plate boundaries
- Earthquakes
- All About Volcanoes
- Geologic Time Scale and Important Geologic Events
- Hydrological Cycle
- The Big Bang Theory
- The Solar System
- The Formation of Earth
- The Life Cycle of a Star
- Types of Eclipses
Don’t just stop at videos or this reviewer. Check out the books and websites in the reference section of this article to get more in-depth information on Earth science topics. Check out our downloadable flashcards and worksheets to help you ace the Earth science portion of the UPCAT and other major examinations.
References
IAU. (n.d.). Retrieved September 02, 2020, from https://www.iau.org/news/pressreleases/detail/iau0603/
NASA Earth Science. (n.d.). Retrieved August 19, 2020, from https://science.nasa.gov/earth-science
NASA Solar System Exploration. (2018, August 31). Retrieved September 02, 2020, from https://solarsystem.nasa.gov/
Reyes, P. J., Bornas, M. A., Dominey-Howes, D., Pidlaoan, A. C., Magill, C. R., & Solidum, J. R. (2018). A synthesis and review of historical eruptions at Taal Volcano, Southern Luzon, Philippines. Earth-Science Reviews, 177, 565-588. doi:10.1016/j.earscirev.2017.11.014
Saugstad, O. D., Oei, J., Lakshminrusimha, S., & Vento, M. (2018). Oxygen therapy of the newborn from molecular understanding to clinical practice. Pediatric Research, 85(1), 20-29. doi:10.1038/s41390-018-0176-8
Sharp, T. (2017). How Big Is Earth?. Retrieved 20 August 2020, from https://www.space.com/17638-how-big-is-earth.html
Tarbuck, Edward J. Lutgens, Frederick K. Earth Science. Upper Saddle River, N.J.: Prentice Hall/Pearson, 2012.
Volcanoes of the Philippines. Retrieved 22 August 2020, from https://www.phivolcs.dost.gov.ph/index.php/volcano-hazard/volcanoes-of-the-philippines
Wicander, R., & Monroe, J. S. (2016). The Dynamic and Evolving Earth. In Historical geology (pp. 1-9). Australia: Cengage Learning.
Written by Ruth Raganit
Ruth Raganit
Ruth Raganit obtained her Bachelor of Science degree in Geology from the University of the Philippines – Diliman. Her love affair with Earth sciences began when she saw a pretty rock and wondered how it came to be. She also likes playing video games, doing digital art, and reading manga.
Copyright Notice
All materials contained on this site are protected by the Republic of the Philippines copyright law and may not be reproduced, distributed, transmitted, displayed, published, or broadcast without the prior written permission of filipiknow.net or in the case of third party materials, the owner of that content. You may not alter or remove any trademark, copyright, or other notice from copies of the content. Be warned that we have already reported and helped terminate several websites and YouTube channels for blatantly stealing our content. If you wish to use filipiknow.net content for commercial purposes, such as for content syndication, etc., please contact us at legal(at)filipiknow(dot)net